The first law of thermodynamics can be written as $ \Delta U = \Delta Q + \Delta W$ for an ideal gas. Which of the following statements is correct?
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas expands adiabatically at constant pressure such that its temperature $T \propto \frac{1}{{\sqrt V }}$, the value of ${C_P}/{C_V}$ of gas isView Solution
- 2A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
- 3View SolutionThe isothermal bulk modulus of a perfect gas at normal pressure is
- 4Three different processes that can occur in an ideal monoatomic gas are shown in the $P$ vs $V$ diagram. The paths are labelled as $A \rightarrow B, A \rightarrow C$ and $A \rightarrow D .$ The change in internal energies during these process are taken as $E _{ AB }, E _{ AC }$ and $E _{ AD }$ and the work done as $W _{ AB }$ $W _{ AC }$ and $W _{ AD }$View Solution
The correct relation between these parameters are
- 5View SolutionWhich statement is incorrect?
- 6For an isothermal expansion of a perfect gas, the value of $\frac{{\Delta P}}{P}$ is equalView Solution
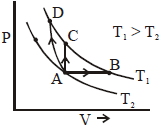
- 7View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
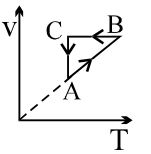
- 8A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
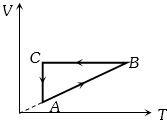
- 9An ideal system can be brought from state $A$ to $B$ through four paths as shown in the figure. The energy given to the system is minimum inView Solution
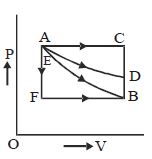
- 10A closed container contains a homogeneous mixture of two moles of an ideal monatomic gas $(\gamma=5 / 3)$ and one mole of an ideal diatomic gas $(\gamma=7 / 5)$. Here, $\gamma$ is the ratio of the specific heats at constant pressure and constant volume of an ideal gas. The gas mixture does a work of $66$ Joule when heated at constant pressure. The change in its internal energy is. . . . . . .Joule.View Solution
