A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)
Medium
$\mathrm{PV}^{\gamma}=$ constant (Adiabatic suddenly)
$\mathrm{P}_{1} \mathrm{V}_{1}^{\gamma}=\mathrm{P}_{2} \mathrm{V}_{2}^{\gamma}$
or $\quad \mathrm{T} \mathrm{P}^{(1-\gamma) / \gamma}=$ constant
$\mathrm{T}_{1} \mathrm{P}_{1}^{(1-\mathrm{\gamma}) / \gamma}=\mathrm{T}_{2} \mathrm{P}_{2}^{(1-\gamma) / \gamma}$
$\mathrm{T}_{2}=\mathrm{T}_{1}\left(\frac{\mathrm{P}_{1}}{\mathrm{P}_{2}}\right)^{(\mathrm{1}-\mathrm{y}) / \gamma}=\mathrm{T}_{1}(2)^{\frac{-0.4}{1.4}}=\frac{300}{(4)^{1 / 7}}$
$\Rightarrow \mathrm{T}_{2}=246 \mathrm{K}=-27^{\circ} \mathrm{C}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
- 2Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
- 3If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
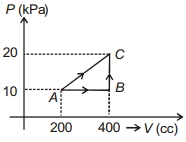
- 4View SolutionIf a gas is heated at constant pressure, its isothermal compressibility
- 5A Carnot engine with efficiency $50\,\%$ takes heat from a source at $600\,K$. In order to increase the efficiency to $70\,\%$, keeping the temperature of sink same, the new temperature of the source will be $.........\,K$View Solution
- 6View SolutionTwo gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally
- 7A cyclic process for $1\,mole$ of an ideal gas is shown. Find work done in $AB, BC$ and $CA$ respectivelyView Solution
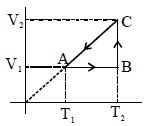
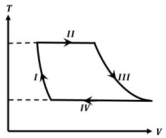
- 8One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (where $V$ is the volume and $T$ is the temperature). Which of the statements below is (are) true?View Solution
(image)
$(A)$ Process $I$ is an isochoric process $(B)$ In process $II$, gas absorbs heat
$(C)$ In process $IV$, gas releases heat $(D)$ Processes $I$ and $III$ are $not$ isobaric
- 9A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.View Solution
- 10A vessel containing $5\, litres$ of a gas at $0.8 \,pa$ pressure is connected to an evacuated vessel of volume $3$ litres. The resultant pressure inside will be ...... $pa$ (assuming whole system to be isolated)View Solution
