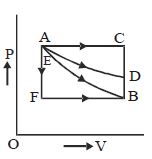
An ideal system can be brought from state $A$ to $B$ through four paths as shown in the figure. The energy given to the system is minimum in

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
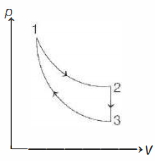
- 1The three processes in a thermodynamic cycle shown in the figure are : Process $1 \rightarrow 2$ is isothermal; Process $2 \rightarrow 3$ is isochoric (volume remains constant); Process $3 \rightarrow 1$ is adiabatic. The total work done by the ideal gas in this cycle is $10 \,J$. The internal energy decreases by $20 \,J$ in the isochoric process. The work done by the gas in the adiabatic process is $-20 \,J$. The heat added to the system in the isothermal process is .............. $J$View Solution
- 2Two Carnot engines $A$ and $B$ are operated in series. Engine $A$ receives heat from a reservoir at $600\,K$ and rejects heat to a reservoir at temperature $T$. Engine $B$ receives; heat rejected by engine $A$ and in turn rejects it to a reservoir at $100\,K$. If the efficiencies of the two engines $A$ and $B$ are represented by ${\eta _A}$ and ${\eta _B}$ respectively, then what is the value of $\frac{{{\eta _A}}}{{{\eta _B}}}$View Solution
- 3$N _{2}$ gas is heated from $300\, K$ temperature to $600\, K$ through an isobaric process. Then find the change in entropy of the gas. $( n =1 mole )$ (in $J/K$)View Solution
- 4One mole of an ideal monoatomic gas is heated at a constant pressure of one atmosphere from ${0^o}C$ to ${100^o}C$. Then the change in the internal energy isView Solution
- 5If $R =$ universal gas constant, the amount of heat needed to raise the temperature of $2$ mole of an ideal monoatomic gas from $273K$ to $373K$ when no work is done ...... $R$View Solution
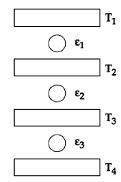
- 6Three Carnot engines operate in series between a heat source at a temperature $T_1$ and a heat sink at temperature $T_4$ (see figure). There are two other reservoirs at temperature $T_2$ and $T_3$, as shown, with $T_1 > T_2 > T_3 > T_4$. The three engines are equally efficient ifView Solution
- 7Given below are two statementView Solution
Statement $-I$ : What $\mu$ amount of an ideal gas undergoes adiabatic change from state $\left( P _{1}, V _{1}, T _{1}\right)$ to state $\left( P _{2}, V _{2}, T _{2}\right)$, the work done is $W =\frac{1 R \left( T _{2}- T _{1}\right)}{1-\gamma}$, where $\gamma=\frac{ C _{ P }}{ C _{ V }}$ and $R =$ universal gas constant,
Statement $-II$ : In the above case. when work is done on the gas. the temperature of the gas would rise.
Choose the correct answer from the options given below
- 8Two identical containers $A$ and $B$ with frictionless pistons contain the same ideal gas at the same temperature and the same volume $V$. The mass of the gas in $A$ is ${m_A}$ and that in $B$ is ${m_B}$. The gas in each cylinder is now allowed to expand isothermally to the same final volume $2V$. The changes in the pressure in $A$ and $B$ are found to be $\Delta P$ and $1.5 \Delta P$ respectively. ThenView Solution
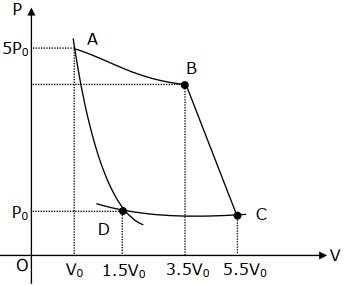
- 9In the reported figure, there is a cyclic process $ABCDA$ on a sample of $1\, {mol}$ of a diatomic gas. The temperature of the gas during the process ${A} \rightarrow {B}$ and ${C} \rightarrow {D}$ are ${T}_{1}$ and ${T}_{2}\left({T}_{1}\,>\,{T}_{2}\right)$ respectively.View Solution
Choose the correct option out of the following for work done if processes $B C$ and $D A$ are adiabatic.
- 10An ideal refrigerator has a freezer at a temperature of $ - 13^\circ C.$ The coefficient of performance of the engine is $5.$ The temperature of the air (to which heat is rejected) will beView Solution
