The 'Kangri' is an earthen pot used to stay warm in Kashmir during the winter months. Assume that the 'Kangri' is spherical and of surface area $7 \times 10^{-2} \,m ^{2}$. It contains $300 g$ of a mixture of coal, wood and leaves with calorific value of $30 \,kJ / g$ (and provides heat with $10 \%$ efficiency). The surface temperature of the 'Kangri' is $60^{\circ} C$ and the room temperature is $0^{\circ} C$. Then, a reasonable estimate for the duration $t$ (in h) that the 'Kangri' heat will last is (take the 'Kangri' to be a black body)
KVPY 2020, Advanced
$(b)$ Given, $A=7 \times 10^{-2} \,m ^{2}$
$m=300 \,g$
Calorific value $=30 \,kJ / g$
Efficiency, $\eta=10 \%=0.1$
$T_{1}=0^{\circ} C =273 \,K$
$T_{2}=60^{\circ} C =333 \,K$
$e=1$ (black body)
From Newton's law of cooling,
$\frac{d Q}{d t}=e A \sigma\left(T_{2}^{4}-T_{1}^{4}\right)$
$=1 \times 7 \times 10^{2} \times 5.735 \times 10^{8}$
$=26.75 \,W$
$\text { Total heat produced, }$
$H =\eta m \times \text { calorific value }$
$\left.=0.1 \times 300 \times 30 \times 10^{3}-(273)^{4}\right]$
$=9 \times 10^{5} \,J$
$\therefore \text { Time, }$ $t =\frac{\text { Heat produced }(H)}{\text { Rate of emission }\left(\frac{d Q}{d t}\right)}$
$=\frac{9 \times 10^{5}}{26.75 \times 3600}=9.35 \,h \simeq 10 \,h$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider two hot bodies ${B_1}$ and ${B_2}$ which have temperatures ${100^o}C$ and ${80^o}C$ respectively at $t = 0$. The temperature of the surroundings is ${40^o}C$. The ratio of the respective rates of cooling ${R_1}$ and ${R_2}$ of these two bodies at $t = 0$ will beView Solution
- 2A black coloured solid sphere of radius $R$ and mass $M$ is inside a cavity with vacuum inside. The walls of the cavity are maintained at temperature $T_0$. The initial temperature of the sphere is $3T_0$. If the specific heat of the material of the sphere varies as $\alpha T^3$ per unit mass with the temperature $T$ of the sphere, where $\alpha $ is a constant, then the time taken for the sphere to cool down to temperature $2T_0$ will be ( $\sigma $ is Stefan Boltzmann constant)View Solution
- 3Two rods of same length and material transfer a given amount of heat in $12$ seconds, when they are joined end to end. But when they are joined lengthwise, then they will transfer same heat in same conditions in ....... $\sec$View Solution
- 4View SolutionThere is a black spot on a body. If the body is heated and carried in dark room then it glows more. This can be explained on the basis of
- 5View SolutionIn variable state, the rate of flow of heat is controlled by
- 6Two stars emit maximum radiation at wavelength $3600Å$ and $4800Å$ respectively. The ratio of their temperatures isView Solution
- 7View SolutionThe value of Stefan’s constant is
- 8A rod of length $L$ with sides fully insulated is of a material whose thermal conductivity varies with $\alpha$ temperature as $ K= \frac{\alpha }{T}$, where $\alpha$ is a constant. The ends of the rod are kept at temperature $T_1$ and $T_2$. The temperature $T$ at $x,$ where $x$ is the distance from the end whose temperature is $T_1$ isView Solution
- 9Four rods of same material and having the same cross section and length have been joined, as shown. The temperature of the junction of four rods will be ............... $^{\circ} C$View Solution

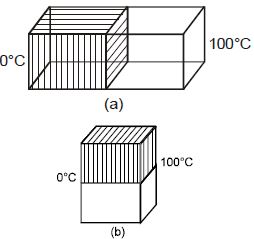
- 10Two identical square rods of metal are welded end to end as shown in figure $(a)$. Assume that $10\, cal$ of heat flows through the rods in $2\, min$. Now the rods are welded as shown in figure, $(b)$. The time it would take for $10$ cal to flow through the rods now, is ........ $\min$View Solution