The pressure and temperature of two different gases is $P$ and $T$ having the volume $V$ for each. They are mixed keeping the same volume and temperature, the pressure of the mixture will be
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At what temperature volume of an ideal gas at $0°C$ becomes triple ....... $^oC$View Solution
- 2The root mean square speed of molecules of nitrogen gas at $27^{\circ} C$ is approximately$.......m/s$(Given mass of a nitrogen molecule $=4.6 \times 10^{-26}\,kg$ and take Boltzmann constant $k _{ B }=1.4 \times 10^{-23}\,JK ^{-1}$ )View Solution
- 3A gaseous mixture contains equal number of hydrogen and nitrogen molecules. Specific heat measurements on this mixture at temperatures below $100\, K$ would indicate that the value of $\gamma $ (ratio of specific heats) for this mixture isView Solution
- 4The temperature at which root mean square speed of a gas will be half of its value at $0\,^oC$ is ....... $^oC$View Solution
- 5A cylinder of fixed capacity of $44.8 \,litres$ contains helium gas at standard temperature and pressure. The amount of heat needed to raise the temperature of gas in the cylinder by $20.0^{\circ} C$ will be .............. $J$(Given gas constant $R =8.3 \,JK ^{-1}- moI ^{-1}$ )View Solution
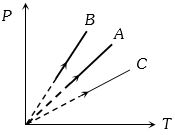
- 6Pressure versus temperature graph of an ideal gas at constant volume $V$ of an ideal gas is shown by the straight line $A$. Now mass of the gas is doubled and the volume is halved, then the corresponding pressure versus temperature graph will be shown by the lineView Solution
- 7An engineer is given a fixed volume $V_m$ of metal with which to construct a spherical pressure vessel. Interestingly, assuming the vessel has thin walls and is always pressurized to near its bursting point, the amount of gas the vessel can contain, $n$ (measured in moles), does not depend on the radius $r$ of the vessel : instead it depends only on $V_m$ (measured in $m^3$) the temperature $T$ (mensured in $K$). the ideal gas constant $R$ (measured in $J/(K\ mol$ )), and the tensile strength of the metal $\sigma $ (measured in $N/m^2$ ) . Which of the following gives $n$ in terms of these parameters?View Solution
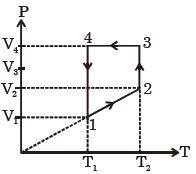
- 8A monoatomic ideal gas of two moles is taken through a cyclic process starting from $1$ as shown. $\frac{{{V_2}}}{{{V_1}}} = 2$ and $\frac{{{V_4}}}{{{V_1}}} = 4$ and temperature iast : $1$ is $T_1 = 27\,^oC$. The temperature at $2$ i.e., $T_2$ ...... $K$View Solution
- 9View SolutionFour mole of hydrogen, two mole of helium and one mole of water vapour form an ideal gas mixture. What is the molar specific heat at constant pressure of mixture?
- 10View SolutionThe parameter that remains the same for molecules of all gases at a given temperature is :
