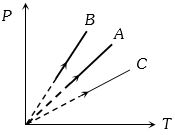
Pressure versus temperature graph of an ideal gas at constant volume $V$ of an ideal gas is shown by the straight line $A$. Now mass of the gas is doubled and the volume is halved, then the corresponding pressure versus temperature graph will be shown by the line

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At which temperature the r.m.s. velocity of a hydrogen molecule equal to that of an oxygen molecule at $47^{\circ} \mathrm{C}$ ?View Solution
- 2A cylindrical container of volume $4.0 \times 10^{-3} \,{m}^{3}$ contains one mole of hydrogen and two moles of carbon dioxide. Assume the temperature of the mixture is $400 \,{K}$ The pressure of the mixture of gases is:View Solution
[Take gas constant as $8.3\, {J} {mol}^{-1} {K}^{-1}$]
- 3Volume-temperature graph at atmospheric pressure for a monoatomic gas $(V\,{\rm{in }}{m^3},\,\,T\,{\rm{in}}{{\rm{ }}^{\rm{o}}}C)$ isView Solution
- 4The mean free path of molecules ofa gas, (radius $r$) is inversely proportional toView Solution
- 5For a gas the difference between the two specific heats is $4150\, J/kg\, K.$ What is the specific heats at constant volume of gas if the ratio of specific heat is $1.4$View Solution
- 6The volume of a gas at $20°C$ is $200\, ml$. If the temperature is reduced to $-20°C$ at constant pressure, its volume will be ...... $ml$View Solution
- 7A gas mixture consists of $3$ moles of oxygen and $5$ moles of argon at temperature $T$. Assuming the gases to be ideal and the oxygen bond to be rigid, the total internal energy (in units of $RT$ ) of the mixture isView Solution
- 8The kinetic energy of translation of $20\, gm$ of oxygen at $47°C$ is (molecular wt. of oxygen is $32 \,gm/mol$ and $R = 8.3\, J/mol/K)$View Solution
- 9A very tall vertical cylinder is filled with a gas of molar mass $M$ under isothermal conditions at temperature $T.$ The density and pressure of the gas at the base of the container is $\rho_0$ and $p_0$, respectively Choose the correct statement(s)View Solution
- 10$Assertion :$ At a given temperature the specific heat of a gas at constant pressure. is always greater than its specific heat at constant volumeView Solution
$Reason :$ When a gas is heated at constant volume some extra heat is needed compared to that at constant pressure for doing work in expansion.
