The rate of diffusion is
AIIMS 1998, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider the following statements for air molecules in an air tight container :View Solution
$(I)$ The average speed of molecules is larger than root mean square speed.
$(II)$ Mean free path of molecules is larger than the mean distance between molecules.
$(III)$ Mean free path of molecules increases with temperature.
$(IV)$ The rms speed of nitrogen is smaller than oxygen molecule.
Which of the above statements are correct?
- 2The average translational kinetic energy of a hydrogen gas molecules at $NTP$ will beView Solution
[Boltzmann’s constant ${k_B} = 1.38 \times {10^{ - 23}}J/K]$
- 3The capacity of a vessel is $3$ litres. It contains $6 \,gm$ oxygen, $8\, gm$ nitrogen and $5\, gm$ $C{O_2}$ mixture at $27°C.$ If $R = 8.31\, J/mole$ $ \times $ $kelvin,$ then the pressure in the vessel in $N/{m^2}$ will be (approx.)View Solution
- 4View SolutionWhich law states that effect of pressure is same for all portion
- 5A box contains n molecules of a gas. How will the pressure of the gas be effected, if the number of molecules is made $2n$View Solution
- 6The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
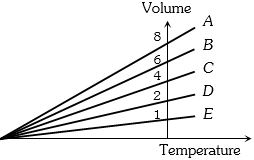
- 7The molar specific heat of a gas as given from the kinetic theory is $\frac{5}{2} R$. If it is not specified whether it is $C _{ P }$ or $C _{ V }$, one could conclude that the molecules of the gasView Solution
- 8View SolutionFour mole of hydrogen, two mole of helium and one mole of water vapour form an ideal gas mixture. What is the molar specific heat at constant pressure of mixture?
- 9The translatory kinetic energy of a gas per $gm$ isView Solution
- 10The pressure of an ideal gas is written as $E = \frac{3PV}{2}$ . Here $E$ stands forView Solution
