The translatory kinetic energy of a gas per $gm$ is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The ratio of specific heats $(\gamma )$ of a ideal gas is given byView Solution
- 2The temperature at which the average translational kinetic energy of a molecule is equal to the energy gained by an electron in accelerating from rest through a potential difference of $1\, volt$ isView Solution
- 3A balloon contains $500\,{m^3}$ of helium at $27°C$ and $1$ atmosphere pressure. The volume of the helium at $-3°C$ temperature and $0.5$ atmosphere pressure will be ..... $m^3$View Solution
- 4View SolutionIf the mean free path of atoms is doubled then the pressure of gas will become
- 5The kinetic energy of one mole gas at $300K$ temperature, is $E.$ At $400K$ temperature kinetic energy is $E'.$ The value of $E'/E$ isView Solution
- 6At which of the following temperature would the molecules of a gas have twice the average kinetic energy they have at $20°C$View Solution
- 7The equation of state of a gas is given by $\left( {P + \frac{{a{T^2}}}{V}} \right)\,{V^c} = (RT + b)$, where $a, b, c$ and $R$ are constants. The isotherms can be represented by $P = A{V^m} - B{V^n}$, where $A$ and $B$ depend only on temperature thenView Solution
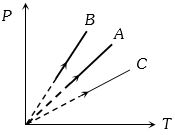
- 8Pressure versus temperature graph of an ideal gas at constant volume $V$ of an ideal gas is shown by the straight line $A$. Now mass of the gas is doubled and the volume is halved, then the corresponding pressure versus temperature graph will be shown by the lineView Solution
- 9An ideal gas $(\gamma = 1.5)$ is expanded adiabatically. How many times has the gas to be expanded to reduce the root mean square velocity of molecules $2.0$ timesView Solution
- 10An ideal gas in a closed container is slowly heated. As its temperature increases, which of the following statements are true$?$View Solution
$(A)$ the mean free path of the molecules decreases.
$(B)$ the mean collision time between the molecules decreases.
$(C)$ the mean free path remains unchanged.
$(D)$ the mean collision time remains unchanged.
