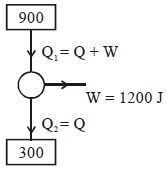
A Carnot engine operates between two reservoirs of temperatures $900\; \mathrm{K}$ and $300 \;\mathrm{K}$ The engine performs $1200\; \mathrm{J}$ of work per cycle. The heat energy (in $\mathrm{J}$ ) delivered by the engine to the low temperature reservoir, in a cycle. is
JEE MAIN 2020, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionTwo gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally, if the two gases are compressed to the same final volume
- 2The isothermal Bulk modulus of an ideal gas at pressure $P$ isView Solution
- 3View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
- 4View SolutionIn adiabatic expansion
- 5An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$View Solution
- 6The internal energy change in a system that has absorbed $2 \;k cal$ of heat and done $500 \;J $ of work is ...... $J$View Solution
- 7$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
$Reason :$ Entropy increases in all natural processes. - 8A Carnot engine take $5000 \,k\,cal$ of heat from a reservoir at $727\,^{\circ}C$ and gives heat to a $\operatorname{sink}$ at $127\,^{\circ}C$. The work done by the engine is $.......... \times 10^{6}\,J$View Solution
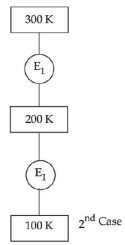
- 9In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.View Solution
- 10A ideal monoatomic gas is carried around the cycle $ABCDA$ as shown in the fig. The efficiency of the gas cycle isView Solution