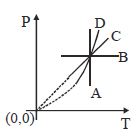
Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Work done by a system under isothermal change from a volume ${V_1}$ to ${V_2}$ for a gas which obeys Vander Waal's equation $(V - \beta n)\,\left( {P + \frac{{\alpha {n^2}}}{V}} \right) = nRT$View Solution
- 2An engine takes in $5$ moles of air at $20\,^{\circ} C$ and $1$ $atm,$ and compresses it adiabaticaly to $1 / 10^{\text {th }}$ of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be $X\, kJ$. The value of $X$ to the nearest integer isView Solution
- 3$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
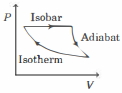
$Reason :$ Entropy increases in all natural processes. - 4The efficiency of the cycle shown below in the figure (consisting of one isobar, one adiabat and one isotherm) is $50 \%$. The ratio $x$, between the highest and lowest temperatures attained in this cycle obeys (the working substance is an ideal gas)View Solution
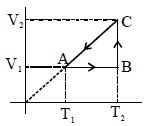
- 5A cyclic process for $1\,mole$ of an ideal gas is shown. Find work done in $AB, BC$ and $CA$ respectivelyView Solution
- 6An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
- 7A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle isView Solution
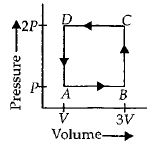
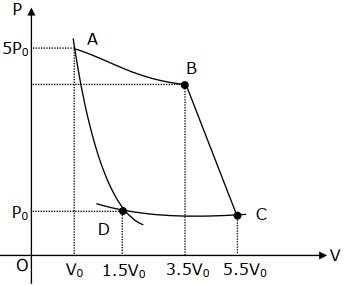
- 8In the reported figure, there is a cyclic process $ABCDA$ on a sample of $1\, {mol}$ of a diatomic gas. The temperature of the gas during the process ${A} \rightarrow {B}$ and ${C} \rightarrow {D}$ are ${T}_{1}$ and ${T}_{2}\left({T}_{1}\,>\,{T}_{2}\right)$ respectively.View Solution
Choose the correct option out of the following for work done if processes $B C$ and $D A$ are adiabatic.
- 9A Carnot engine has efficiency of $50 \%$. If the temperature of sink is reduced by $40^{\circ} C$, its efficiency increases by $30 \%$. The temperature of the source will be$....K$View Solution
- 10A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
