$40$ calories of heat is needed to raise the temperature of $1\, mole$ of an ideal monoatomic gas from $20°C$ to $30°C$ at a constant pressure. The amount of heat required to raise its temperature over the same interval at a constant volume $(R = 2\,calorie\,mol{e^{ - 1}}{K^{ - 1}})$ is ..... $calorie$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$r.m.s.$ velocity of nitrogen molecules at $NTP$ is ...... $m/s$View Solution
- 2$P-T$ diagram of an ideal gas having three different densities $\rho_1, \rho_2, \rho_3$ (in three different cases) is shown in the figure. Which of the following is correct :View Solution
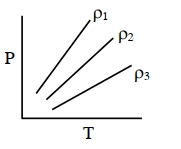
- 3The volume occupied by the molecules contained in $4.5\,kg$ water at $STP$, if the intermolecular forces vanish away is ........ $m ^{3}$View Solution
- 4An inverted bell lying at the bottom of a lake $47.6\,m$ deep has $50\, cm^3$ of air trapped in it. The bell is brought to the surface of the lake. The volume of the trapped air will be ...... $cm^3$ (atmospheric pressure $= 70\, cm$ of $Hg$ and density of $Hg = 13.6\, g/cm^3$)View Solution
- 5Given below are two statements:View Solution
Statements $I:$ The temperature of a gas is $-73^{\circ}\,C$. When the gas is heated to $527^{\circ}\,C$, the root mean square speed of the molecules is doubled.
Statement $II:$ The product of pressure and volume of an ideal gas will be equal to translational kinetic energy of the molecules.
In the light of the above statements, choose the correct answer from the options given below :
- 6In the two vessels of same volume, atomic hydrogen and helium at pressure $1\, atm$ and $2\, atm$ are filled. If temperature of both the samples is same, then average speed of hydrogen atoms $ < {C_H} > $ will be related to that of helium $ < {C_{He}} > $ asView Solution
- 7The specific heats, $C_P$ and $C_V$ of a gas of diatomic molecules, $A$, are given (in units of $J\, mol^{-1}\, K^{-1}$) by $29$ and $22$, respectively. Another gas of diatomic molecules $B$, has the corresponding values $30$ and $21$. If they are treated as ideal gases, thenView Solution
- 8Graph between volume and temperature for a gas is shown in figure. If $\alpha$ = Volume coefficient of gas = $\frac{1}{273}$ per $^o C$,then what is the volume of gas at a temperature of $819 ^o C$View Solution
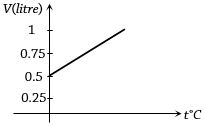
- 9One mole of an ideal monatomic gas requires $210 \,J$ heat to raise the temperature by $10\, K$, when heated at constant temperature. If the same gas is heated at constant volume to raise the temperature by $10\, K$ then heat required is ....... $J$View Solution
- 10A flask contains argon and oxygen in the ratio of $3: 2$ in mass and the mixture is kept at $27^{\circ} C$. The ratio of their average kinetic energy per molecule respectively will be ...........View Solution
