The volume occupied by the molecules contained in $4.5\,kg$ water at $STP$, if the intermolecular forces vanish away is ........ $m ^{3}$
NEET 2022, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1We write the relation for Boyle's law in the form $PV = C$ when the temperature remains constant. In this relation, the magnitude of $C$ depends uponView Solution
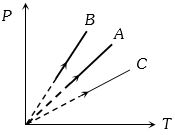
- 2Pressure versus temperature graph of an ideal gas at constant volume $V$ of an ideal gas is shown by the straight line $A$. Now mass of the gas is doubled and the volume is halved, then the corresponding pressure versus temperature graph will be shown by the lineView Solution
- 3In gases of diatomic molecules, the ratio of the two specific heats of gases ${C_P}/{C_V}$ isView Solution
- 4The average translational kinetic energy of a hydrogen gas molecules at $NTP$ will beView Solution
[Boltzmann’s constant ${k_B} = 1.38 \times {10^{ - 23}}J/K]$
- 5View SolutionThe specific heat of a gas in a polytropic process is given by
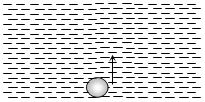
- 6An air bubble doubles its radius on raising from the bottom of water reservoir to be the surface of water in it. If the atmospheric pressure is equal to $10\, m$ of water, the height of water in the reservoir is ..... $m$View Solution
- 7At what temperature will the oxygen molecules have the same root mean square speed as hydrogen molecules at $200 \,K$ ....... $K$View Solution
- 8In Vander Waal's equation $\left[ {P + \frac{a}{{{V^2}}}} \right]\,(V - b) = RT,$ the dimensions of $a$ areView Solution
- 9The $r.m.s.$ speed of the molecules of a gas in a vessel is $400$ $m{s^{ - 1}}$. If half of the gas leaks out, at constant temperature, the $r.m.s.$ speed of the remaining molecules will be ..... $ms^{-1}$View Solution
- 10The average translational kinetic energy of ${O_2}$ (molar mass $32$) molecules at a particular temperature is $ 0.048\, eV.$ The translational kinetic energy of ${N_2}$ (molar mass $28$) molecules in $eV$ at the same temperature isView Solution
