In Vander Waal's equation $\left[ {P + \frac{a}{{{V^2}}}} \right]\,(V - b) = RT,$ the dimensions of $a$ are
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The graph between pressure $(P)$ and $\frac{1}{V}$ ($V$ is volume) for constant temperature process in ideal equation $(PV = nRT)$View Solution
- 2Pressure $Vs$ temperature graph of an ideal gas is shown in figure. Density of the gas at point $A$ is $\rho _0$ , Density at $B$ will beView Solution
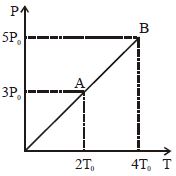
- 3The value closest to the thermal velocity of a Helium atom at room temperature $(300\,K)$in $ms^{-1}$ is $[k_B\, = 1 .4\times10^{-23}\,J/K;\, m_{He}\, = 7\times10^{-27}\,kg]$View Solution
- 4A partition divides a container having insulated walls into two compartments $I$ and $II$. the same gas fills the two compartments. The ratio of the number of molecules in compartments $I$ and $II$ isView Solution

- 5For a certain process, pressure of diatomic gas varies according to the relation $P = aV^2$, where $a$ is constant. What is the molar heat capacity of the gas for this process?View Solution
- 6The root mean square speed of the molecules of a diatomic gas is $v$. When the temperature is doubled, the molecules dissociate into two atoms. The new root mean square speed of the atom isView Solution
- 7A gaseous mixture consists of $16\,g$ of helium and $16\,g$ of oxygen. The ratio $\frac{{{C_P}}}{{{C_V}}}$ of the mixture isView Solution
- 8The number of air molecules in a $(5 \,m \times 5 \,m \times 4 \,m )$ room at standard temperature and pressure is of the order ofView Solution
- 9A pressure cooker contains air at $1$ atm and $30^o C$. If the safety value of the cooler blows when the inside pressure $ \ge 3$ atm, then the maximum temperature of the air, inside the cooker can be .... $^oC$View Solution
- 10To what temperature should the hydrogen at room temperature $(27°C)$ be heated at constant pressure so that the $R.M.S.$ velocity of its molecules becomes double of its previous value ....... $^oC$View Solution
