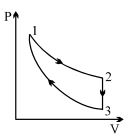
Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$

Advanced
In the process $1 \rightarrow 2, \Delta Q_{1 \rightarrow 2}=\Delta W_{1 \rightarrow 2}$
In the process $2 \rightarrow 3, \Delta W_{2 \rightarrow 3}=0$
In the process $3 \rightarrow 1, \Delta Q_{3 \rightarrow 1}=0$
The complete process being cyclic, $\Delta U=0$
Hence $\Delta Q=\Delta W \Rightarrow \Delta Q_{1 \rightarrow 2}+\Delta Q_{2 \rightarrow 3}+\Delta Q_{3 \rightarrow 1}=\Delta W_{1 \rightarrow 2}+\Delta W_{2 \rightarrow 3}+\Delta W_{3 \rightarrow 1} \Rightarrow$
$\Delta Q_{2 \rightarrow 3}=\Delta W_{3 \rightarrow 1}=-40 J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
- 2One mole of an ideal gas with $\gamma = 1.4$, is adiabatically compressed so that its temperature rises from $27°C$ to $35°C$. The change in the internal energy of the gas is ....... $J$ $(R = 8.3\,J/mol.K)$View Solution
- 3$Assertion :$ In an isolated system the entropy increases.View Solution
$Reason :$ The processes in an isolated system are adiabatic. - 4View SolutionThe adiabatic Bulk modulus of a perfect gas at pressure is given by
- 5A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution
- 6$0.02\, moles$ of an ideal diatomic gas with initial temperature $20^{\circ} C$ is compressed from $1500 \,cm ^{3}$ to $500 \,cm ^{3}$. The thermodynamic process is such that $p V^{2}=\beta$, where $\beta$ is a constant. Then, the value of $\beta$ is close to (the gas constant, $R=8.31 \,J / K / mol$ ).View Solution
- 7If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
- 8Agas expands such that its initial and final temperature are equal. Also, the process followed by the gas traces a straight line on the $P-V$ diagram :View Solution
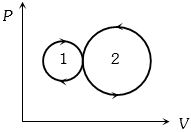
- 9View SolutionIn the following indicator diagram, the net amount of work done will be
- 10In an Isobaric process, the work done by a di$-$atomic gas is $10\, J ,$ the heat given to the gas will be (in $J$)View Solution
