$Assertion :$ In an isolated system the entropy increases.
$Reason :$ The processes in an isolated system are adiabatic.
$Reason :$ The processes in an isolated system are adiabatic.
AIIMS 2006, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Heat is supplied to a diatomic gas at constant pressure. The ratio of $\Delta Q\,:\,\Delta U\,:\,\Delta W$ isView Solution
- 2A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an absolute temperature $T$ and rejects heat to a sink at a temperature of $T/3$ . The amount of heat rejected isView Solution
- 3In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution
- 4The value of $\eta$ may lie betweenView Solution
- 5An engine operates by taking a monatomic ideal gas through the cycle shown in the figure. The percentage efficiency of the engine is close to $.......\%$View Solution
- 6A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an abosolute temperature $T$ and rejects heat to a sink at a temperature of $T/3.$ The amount of heat rejected isView Solution
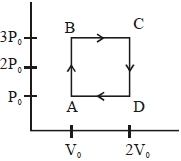
- 7View SolutionIn the following indicator diagram, the net amount of work done will be
- 8A Carnot engine whose heat $\operatorname{sinks}$ at $27\,^{\circ} C$, has an efficiency of $25 \%$. By how many degrees should the temperature of the source be changed to increase the efficiency by $100 \%$ of the original efficiency $?$View Solution
- 9An engine is supposed to operate between two reservoirs at temperature $727°C$ and $227°C.$ The maximum possible efficiency of such an engine isView Solution
- 10One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
