To what temperature should the hydrogen at room temperature $(27°C)$ be heated at constant pressure so that the $R.M.S.$ velocity of its molecules becomes double of its previous value ....... $^oC$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A diatomic gas of molecules weight $30\,\, gm/mole$ is filled in a container at $27\,^oC$. It is moving at a velocity $100\,\, m/s$. If it is suddenly stopped, the rise in temperature of gas is :View Solution
- 2If the ratio of vapour density for hydrogen and oxygen is $\frac{1}{{16}}$, then under constant pressure the ratio of their rms velocities will beView Solution
- 3The figure shows the volume $V$ versus temperature $T$ graphs for a certain mass of a perfect gas at two constant pressures of ${P_1}$ and ${P_2}$. What interference can you draw from the graphsView Solution
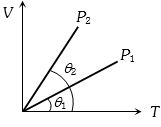
- 4When the temperature of a gas is raised from $27^o C$ to $90^o C$, the percentage increase in the $r.m.s.$ velocity of the molecules will be ..... $\%$View Solution
- 5$2\, moles$ of helium gas is mixed with $3\, moles$ of hydrogen molecules (taken to be rigid). What is the molar specific heat of mixture at constant volume ....... $J/mol\, K$ ? $(R = 8.3\, J/mol\, K)$View Solution
- 6Consider two ideal diatomic gases $\mathrm{A}$ and $\mathrm{B}$ at some temperature $T$. Molecules of the gas $A$ are rigid, and have a mass $m$. Molecules of the gas $\mathrm{B}$ have an additional vibrational mode, and have a mass $\frac{\mathrm{m}}{4} .$ The ratio of the specific heats $(\mathrm{C}_{\mathrm{v}}^{\mathrm{A}}$ and $\mathrm{C}_{\mathrm{v}}^{\mathrm{B}})$ of gas $\mathrm{A}$ and $\mathrm{B}$, respectively isView Solution
- 7View SolutionIf the pressure in a closed vessel is reduced by drawing out some gas, the mean free path of the molecules
- 8For a gas $\gamma = 7/5.$ The gas may probably beView Solution
- 9View SolutionVapour is injected at a uniform rate in a closed vessel which was initially evacuated. The pressure in the vessel
- 10$N( < 100)$ molecules of a gas have velocities $1, 2, 3\,.\,.\,.\,.\,.N/km/s$ respectively. ThenView Solution
