A diatomic gas of molecules weight $30\,\, gm/mole$ is filled in a container at $27\,^oC$. It is moving at a velocity $100\,\, m/s$. If it is suddenly stopped, the rise in temperature of gas is :
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$105$ calories of heat is required to raise the temperature of $3$ moles of an ideal gas at constant pressure from $30^{\circ} C$ to $35^{\circ} C$. The amount of heat required in calories to raise the temperature of the gas through the range $\left(60^{\circ} C\right.$ to $\left.65^{\circ} C \right)$ at constant volume is ........ $cal$ $\left(\gamma=\frac{C_p}{C_v}=1.4\right)$View Solution
- 2View SolutionInternal energy of a non-ideal gas depends on ..........
- 3The relation between root mean square speed $\left( v _{ rms }\right)$ and most probable speed $\left( v _{ p }\right)$ for the molar mass $M$ of oxygen gas molecule at the temperature of $300\, K$ will beView Solution
- 4$Assertion :$ Mean free path of a gas molecules varies inversely as density of the gas.View Solution
$Reason :$ Mean free path varies inversely as pressure of the gas. - 5A closed vessel contains $8\,gm$ of oxygen and $7\,gm$ of nitrogen. The total pressure is $10\, atm$ at a given temperature. If now oxygen is absorbed by introducing a suitable absorbent the pressure of the remaining gas in atm will beView Solution
- 6Two bulbs of identical volumes connected by a small capillary are initially filled with an ideal gas at temperature $T$. Bulb $2$ is heated to maintain a temperature $2 T$, while bulb $1$ remains at temperature $T$. Assume throughout that the heat conduction by the capillary is negligible. Then, the ratio of final mass of the gas in bulb $2$ to the initial mass of the gas in the same bulb is close toView Solution
- 7View SolutionA gas is filled in the cylinder shown in the figure. The two pistons are joined by a string. If the gas is heated, the pistons will
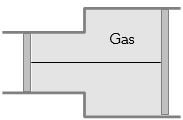
- 8The root mean square speed of molecules of nitrogen gas at $27^{\circ} C$ is approximately$.......m/s$(Given mass of a nitrogen molecule $=4.6 \times 10^{-26}\,kg$ and take Boltzmann constant $k _{ B }=1.4 \times 10^{-23}\,JK ^{-1}$ )View Solution
- 9A vessel contains a mixture of one mole of oxygen and two moles of nitrogen at $300\ K.$ The ratio of the average rotational kinetic energy per $O_2$ molecule to that per $N_2$ molecule isView Solution
- 10View SolutionTwo vessels having equal volume contains molecular hydrogen at one atmosphere and helium at two atmospheres respectively. If both samples are at the same temperature, the mean velocity of hydrogen molecules is
