The time average of the kinetic energy of one molecule of a gas taken over a long period of time
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider a gas with density $\rho $ and $\bar c$ as the root mean square velocity of its molecules contained in a volume. If the system moves as whole with velocity $v,$ then the pressure exerted by the gas isView Solution
- 2The $r.m.s.$ velocity will be greater forView Solution
- 3Which one of the following schematic graphs best represents the variation of $p V$ (in Joules) versus $T$ (in Kelvin) of one mole of an ideal gas? (The dotted line represents $p V=T$ )View Solution
- 4The respective speeds of five molecules are $2, 1.5, 1.6, 1.6$ and $1.2 \,km/sec.$ The most probable speed in $km/sec$ will beView Solution
- 5An $HCl$ molecule has rotational, translational and vibrational motions. If the $rms$ velocity of $HCl$ molecules in its gaseous phase is $\bar v ,\,m$ is its mass and $k_B$ is Boltzmann constant, then its temperature will beView Solution
- 6A vessel contains $16 \,g$ of hydrogen and $128 \,g$ of oxygen at standard temperature and pressure. The volume of the vessel in $cm ^{3}$ isView Solution
- 7The plot that depicts the behavior of the mean free time $t$ (time between two successive collisions) for the molecules of an ideal gas, as a function of temperature $(T)$, qualitatively, is (Graphs are schematic and not drawn to scale)View Solution
- 8A cylinder made of perfectly non conducting material closed at both ends is divided into two equal parts by a heat proof piston. Both parts of the cylinder contain the same masses of a gas at a temperature $t_0 = 27^o$ and pressure $P_0 = 1$ atm. Now if the gas in one of the parts is slowly heated to $t = 57^oC$ while the temperature of first part is maintained at $t_0$ the distance moved by the piston from the middle of the cylinder will be.... $cm$ (length of the cylinder $= 84\,\, cm$)View Solution
- 9From the following $P-T$ graph what interference can be drawnView Solution
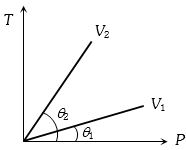
- 10For two different gases $X$ and $Y,$ having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{v_1}$ and $C_{v_2}$ respectively, for adiabatic process , the $\ln P$ versus $\ln V$ graph is plotted as shown :-View Solution