When $2 \,\,gms$ of a gas are introduced into an evacuated flask kept at $25\,^0C$ the pressure is found to be one atmosphere. If $3 \,\,gms$ of another gas added to the same flask the pressure becomes $1.5$ atmospheres. The ratio of the molecular weights of these gases will be
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionVelocity of sound measured at a given temperature in oxygen and hydrogen is in the ratio
- 2View SolutionInternal energy of a non-ideal gas depends on ..........
- 3View SolutionThe specific heat of an ideal gas is
- 4Hydrogen gas and oxygen gas have volume $1 \,\,cm^3$ each at $N.T.P$.View Solution
- 5To raise the temperature of a certain mass of gas by $50^{\circ} C$ at a constant pressure, $160$ calories of heat is required. When the same mass of gas is cooled by $100^{\circ} C$ at constant volume, $240$ calories of heat is released. How many degrees of freedom does each molecule of this gas have (assume gas to be ideal) $?$View Solution
- 6$1$ mole of rigid diatomic gas performs a work of $Q / 5$ when heat $Q$ is supplied to it. The molar heat capacity of the gas during this transformation is $\frac{ x R }{8},$ The value of $x$ is $\ldots \ldots \ldots .$ $[ K =$ universal gas constant $]$View Solution
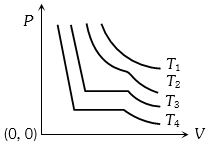
- 7The change in volume $V$ with respect to an increase in pressure $P$ has been shown in the figure for a non-ideal gas at four different temperatures ${T_1},\,{T_2},\,{T_3}$ and ${T_4}$. The critical temperature of the gas isView Solution
- 8The ratio of specific heats $(\gamma )$ of a ideal gas is given byView Solution
- 9The temperature of a gas is $-78^{\circ} \mathrm{C}$ and the average translational kinetic energy of its molecules is $\mathrm{K}$. The temperature at which the average translational kinetic energy of the molecules of the same gas becomes $2 \mathrm{~K}$ is :View Solution
- 10View SolutionFor matter to exist simultaneously in gas and liquid phases
