Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at $T_1 = 800\, K$ and rejects to sink at $T_2K$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at $T_3 = 300\, K$. If the work outputs of two engines are equal, then the value of $T_2$ is ...... $K$
Diffcult
$\eta=\frac{W}{Q}=\frac{T_{1}-T_{2}}{T_{1}}$
$\mathrm{W}_{1}=\mathrm{Q}\left[\frac{800-\mathrm{T}_{2}}{800}\right]$
heat rejected by $A$ is $\mathrm{Q}_{2}=\mathrm{Q}-\mathrm{W}_{1}=\frac{\mathrm{QT}_{2}}{800}$
heat rejected by $1^{\text {st }}=$ input of $2 \mathrm{nd}=\frac{\mathrm{QT}_{2}}{800}$
$\mathrm{W}_{2}=\frac{\mathrm{T}_{2}-300}{\mathrm{T}_{2}} \times \frac{\mathrm{QT}_{2}}{800}=\frac{\mathrm{Q}\left(\mathrm{T}_{2}-300\right)}{800}$
$\mathrm{W}_{1}=\mathrm{W}_{2}$
$Q\left[\frac{800-T_{2}}{800}\right]=\frac{\left(T_{2}-300\right) Q}{800}$
$\Rightarrow 2 \mathrm{T}_{2}=1100 \Rightarrow \mathrm{T}_{2}=550 \mathrm{K}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA measure of the degree of disorder of a system is known as
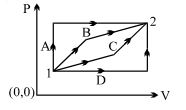
- 2A ideal monoatomic gas is carried around the cycle $ABCDA$ as shown in the fig. The efficiency of the gas cycle isView Solution
- 3A diatomic ideal gas is used in a carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\,V$ , the efficiency of the engine isView Solution
- 4View SolutionWhen an ideal monoatomic gas is heated at constant pressure, fraction of heat energy supplied which increases the internal energy of gas, is
- 5$\Delta U + \Delta W = 0$ is valid forView Solution
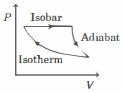
- 6The efficiency of the cycle shown below in the figure (consisting of one isobar, one adiabat and one isotherm) is $50 \%$. The ratio $x$, between the highest and lowest temperatures attained in this cycle obeys (the working substance is an ideal gas)View Solution
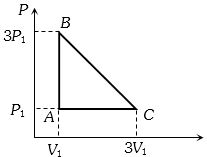
- 7An ideal gas is taken around the cycle $ABCA$ as shown in the $P-V $ diagram. The net work done by the gas during the cycle is equal toView Solution
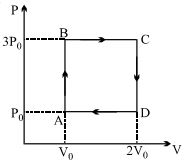
- 8On a $TP$ diagram, two moles of ideal gas perform process $AB$ and $CD$. If the work done by the gas in the process $AB$ is two times the work done in the process $CD$ then what is the value of $T_1/T_2$?View Solution
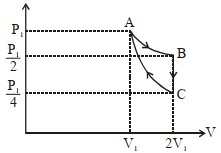
- 9If one mole of an ideal gas at $\left( P _{1}, V _{1}\right)$ is allowed to expand reversibly and isothermally ($A$ to $B$ ) its pressure is reduced to one-half of the original pressure (see $figure$). This is followed by a constant volume cooling till its pressure is reduced to one-fourth of the initial value $( B \rightarrow C ) .$ Then it is restored to its initial state by a reversible adiabatic compression ($C$ to $A$). The net workdone by the gas is equal to ...... .View Solution
- 10An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution