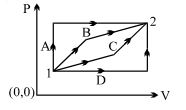
An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. Then

Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The efficiency of carnot engine is $50\%$ and temperature of sink is $500\;K$. If temperature of source is kept constant and its efficiency raised to $60\%$, then the required temperature of the sink will beView Solution
- 2View SolutionTemperature is a measurement of coldness or hotness of an object. This definition is based on
- 3View SolutionThe adiabatic Bulk modulus of a perfect gas at pressure is given by
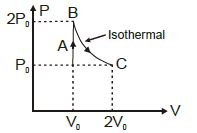
- 4A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution
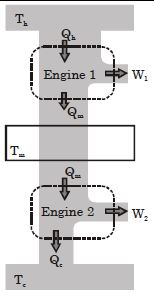
- 5Suppose that two heat engines are connected in series, such that the heat released by the first engine is used as the heat absorbed by the second engine, as shown in figure. The efficiencies of the engines are $\in_1$ and $\in_2$, respectively. The net efficiency of the combination is given by :View Solution
- 6View SolutionIf an ideal gas is compressed isothermally. Which of the following statements is true?
- 7A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion isView Solution
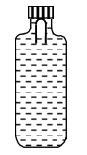
- 8A soft plastic bottle, filled with water of density $1 gm / cc$, carries an inverted glass test-tube with some air (ideal gas) trapped as shown in the figure. The test-tube has a mass of $5 gm$, and it is made of a thick glass of density $2.5 gm / cc$. Initially the bottle is sealed at atmospheric pressure $p_0=10^5 Pa$ so that the volume of the trapped air is $v_0=3.3 cc$. When the bottle is squeezed from outside at constant temperature, the pressure inside rises and the volume of the trapped air reduces. It is found that the test tube begins to sink at pressure $P_0+\Delta p$ without changing its orientation. At this pressure, the volume of the trapped air is $v_0-\Delta v$.View Solution
Let $\Delta v=X$ cc and $\Delta p=Y \times 10^3 Pa$.
($1$) The value of $X$ is
($2$) The value of $Y$ is
Give the answer or quetion ($1$) and ($2$)
- 9Match List $I$ with List $II$ :View Solution
List $I$ List $II$ $A$ Isothermal Process $I$ Work done by the gas decreases internal energy $B$ Adiabatic Process $II$ No change in internal energy $C$ Isochoric Process $III$ The heat absorbed goes partly to increase internal energy and partly to do work $D$ Isobaric Process $IV$ No work is done on or by the gas Choose the correct answer from the options given below :
- 10$100\ g$ of water is heated from $30^o\ C$ to $50^o\ C$ Ignoring the slight expansion of the water, the change in its internal energy is ...... $kJ$ (specific heat of water is $4184\ J/kg/K$)View Solution
