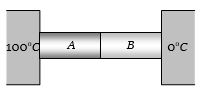
Two metal cubes $A$ and $B$ of same size are arranged as shown in the figure. The extreme ends of the combination are maintained at the indicated temperatures. The arrangement is thermally insulated. The coefficients of thermal conductivity of $A$ and $B$ are $300\;W/m{\;^o}C$ and $200\;W/m{\;^o}C$, respectively. After steady state is reached, the temperature of the interface will be...... $^oC$

IIT 1996, Medium
(d) Temperature of interface $T = \frac{{{K_1}{\theta _1} + {K_2}{\theta _2}}}{{{K_1} + {K_2}}}$
$ = \frac{{300 \times 100 + 200 \times 0}}{{300 + 200}} = 60^\circ C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
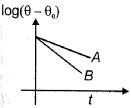
- 1Two bodies $A$ and $B$ of equal masses, area and emissivity cooling under Newton's law of cooling from same temperature are represented by the graph. If $\theta$ is the instantaneous temperature of the body and $\theta_0$ is the temperature of surroundings, then relationship between their specific heats is ..........View Solution
- 2View SolutionIf black wire of platinum is heated, then its colour first appear red, then yellow and finally white. It can be understood on the basis of
- 3View SolutionPick out the statement which is not true
- 4$Assertion :$ Bodies radiate heat at all temperature.View Solution
$Reason :$ Rate of radiation of heat is proportional to the fourth power of absolute temperature. - 5View SolutionThere is a black spot on a body. If the body is heated and carried in dark room then it glows more. This can be explained on the basis of
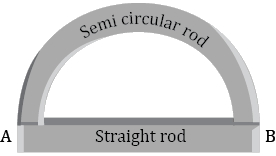
- 6Two rods (one semi-circular and other straight) of same material and of same cross-sectional area are joined as shown in the figure. The points $A$ and $B$ are maintained at different temperature. The ratio of the heat transferred through a cross-section of a semi-circular rod to the heat transferred through a cross section of the straight rod in a given time isView Solution
- 7Liquid is filled in a vessel which is kept in a room with temperature ${20^o}C$. When the temperature of the liquid is ${80^o}C$, then it loses heat at the rate of $60\;cal/\sec $. What will be the rate of loss of heat when the temperature of the liquid is ${40^o}C$ ....... $cal/\sec $View Solution
- 8Inside a closed furnace held at a temperature of $400\,\, K,$ we have a black body. A hole of area $10\,\, cm^2$ is opened in the furnace so that sunlight starts falling on black body. The intensity of sunlight is $2000\, W/m^2.$ In the steady stateView Solution
- 9View SolutionWater and turpentine oil (specific heat less than that of water) are both heated to same temperature. Equal amounts of these placed in identical calorimeters are then left in air
- 10View SolutionIn rainy season, on a clear night the black seat of a bicycle becomes wet because
