Vapours of an alcohol X when passed over hot reduced copper, produce an alkene, the alcohol is:
- Tertiary alcohol
Given: X(ROH) + Cu(hot) → alkene
Primary alcohol passed over Cu at 300°C is dehydrogenated to aldehydes and hydrogen gas liberates.
Secondary alcohols are dehydrogenated to ketones with liberation of hydrogen gas
Tertiary alcohol is dehydrated to alkene.
This method can therefore be used to distinguish between primary, secondary and tertiary alcohol
Thus X should be a tertiary alcohol.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe strongest acid in the following compounds is:
- 2The general formula for alcohol series is $\ce{C_nH_{2n+1}OH}.$ Which is the formula for alcohol contains four carbon?View Solution
- 3View SolutionWhich of the following combinations can be used for the preparation of cis vic diol?
- 4View SolutionWhich of the following cannot be dissolved in alcohol?
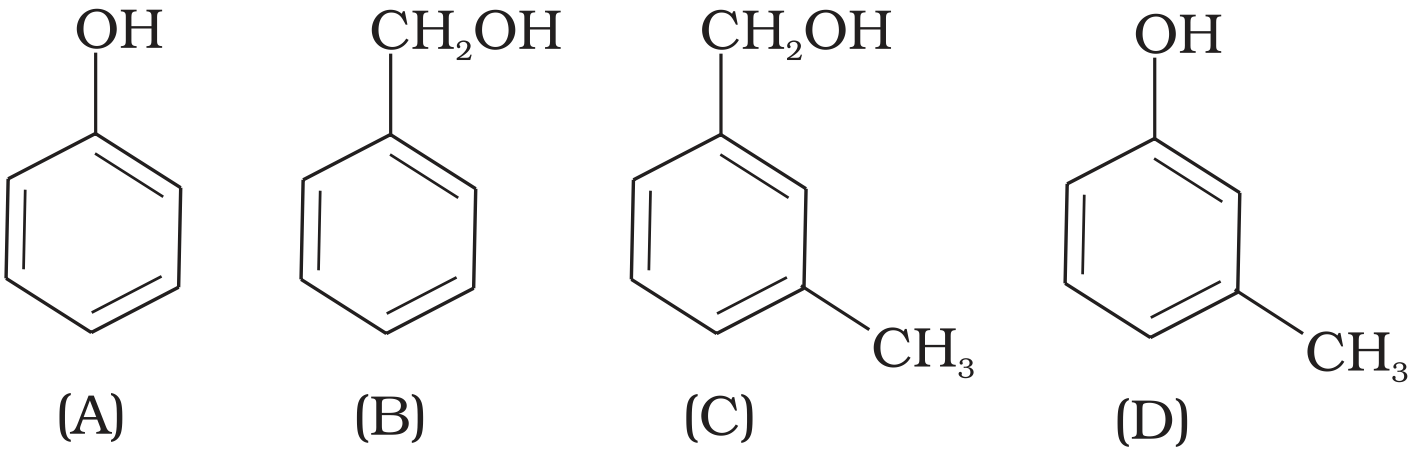
- 5View SolutionWhich of the following compounds is aromatic alcohol?
- 6$\ce{CH_3CH_2OH}$ can be converted into $\ce{CH_3CHO}$ by $.........$View Solution
- 7View SolutionWhich of the following alkenes on acid-catalysed hydration gives a tertiary alcohol:
- 8View SolutionWhich of the following reactions is possible?
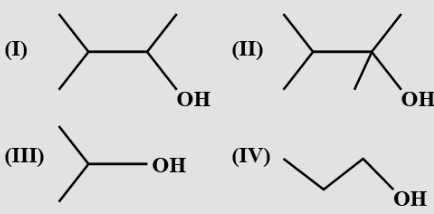
- 9The order of reactivity of the following alcohols towards dehydration is$:$View Solution
- 10View SolutionArrange the following compounds in increasing order of boiling point.
Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol.
