Arrange the following compounds in increasing order of boiling point.
Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol.
Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol.
- Propan-1-ol, butan-2-ol, butan-1-ol, pentan-1-ol
The boiling points of alcohols and phenols increase with increase in the number of carbon atoms (increase in van der Waals forces.). In alcohols, the boiling points decrease with increase of branching in carbon chain (because of decrease in van der Waals forces with decrease in surface area.).
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionMaximum boiling point would be shown by:
- 2View SolutionDestructive distillation of wood in the laboratory will result in the formation of:
- 3Which of the following is produced during the following reaction? $\text{CO(g)} + \text{H}_2\text{(g)}\xrightarrow[\text{ZnO}\text{Cr}_2\text{O}_3]{575\text{k}}$View Solution
- 4IUPAC name of the compound $\text{CH}_3-\text{CH}-\text{OCH}_3 \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{CH}_3$ is ___________.View Solution
- 5The product formed by decolourization of $\ce{KMnO_{4}}$ by ethylene is $.......$ which is used as $.......$View Solution
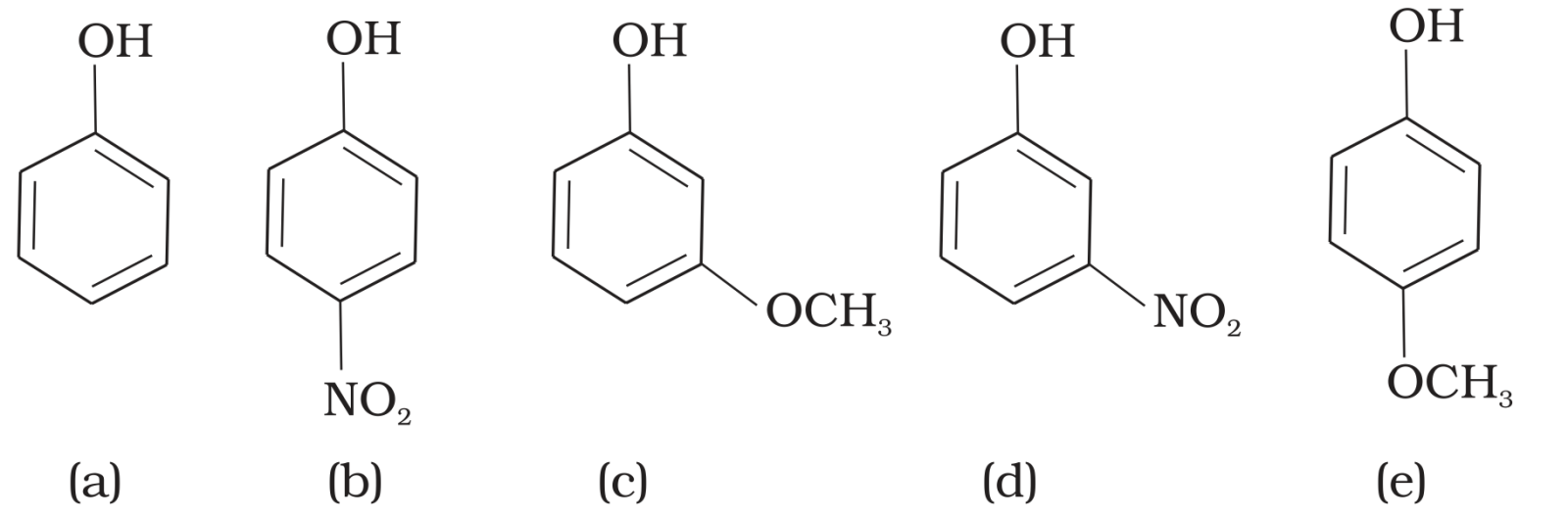
- 6View SolutionStrength of acidity is in order:
- 7The general formula for alcohol series is $\ce{C_nH_{2n+1}OH}.$ Which is the formula for alcohol contains four carbon?View Solution
- 8View SolutionPrimary, secondary and tertiary alcohols can be distinguish by:
- 9View SolutionThe correct order of boiling point for primary (1), secondary (2) and tertiary (3) alcohols is:
- 10View SolutionMark the correct order of decreasing acid strength of the following compounds.