When $1\, kg$ of ice at $0^o C$ melts to water at $0^o C,$ the resulting change in its entropy, taking latent heat of ice to be $80\, cal/gm,$ is ...... $cal/K$
AIPMT 2011, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Carnot engine has an efficiency of $50 \%$ when its source is at a temperature $327^{\circ}\,C$. The temperature of the sink is $.........^{\circ} C$View Solution
- 2View SolutionIrreversible process is
- 3$200\,g$ water is heated from $40\,^oC$ to $60\,^oC.$ Ignoring the slight expansion of water, the change in its internal energy is close to ...... $kJ$ (Given specific heat of water $=4184\,J/kgK$ )View Solution
- 4In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution
- 5A Carnot freezer takes heat from water at $0\,^oC$ inside it and rejects it to the room at a temperature of $27\,^oC$. The latent heat of ice is $336 \times 10^3\, J\,kg^{-1}$. lf $5\, kg$ of water at $0\,^oC$ is converted into ice at $0\,^oC$ by the freezer, then the energy consumed by the freezer is close toView Solution
- 6$Assertion :$ Adiabatic expansion is always accompanied by fall in temperature.View Solution
$Reason :$ In adiabatic process, volume is inversely proportional to temperature. - 7An ideal heat engine working between temperature $T_1$ and $T_2 $ has an efficiency $\eta$, the new efficiency if both the source and sink temperature are doubled, will beView Solution
- 8A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
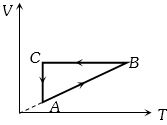
- 9In a carnot engine, the temperature of reservoir is $527^{\circ} C$ and that of $\operatorname{sink}$ is $200 \; K$. If the workdone by the engine when it transfers heat from reservoir to sink is $12000 \; kJ$, the quantity of heat absorbed by the engine from reservoir is $\times 10^{6} \; J$View Solution
- 10$N$ moles of an ideal diatomic gas are in a cylinder at temperature $T$. suppose on supplying heat to the gas, its temperature remain constant but $n$ moles get dissociated into atoms. Heat supplied to the gas isView Solution
