An ideal heat engine working between temperature $T_1$ and $T_2 $ has an efficiency $\eta$, the new efficiency if both the source and sink temperature are doubled, will be
Medium
(b) In first case ${\eta _1} = \frac{{{T_1} - {T_2}}}{{{T_1}}}$
In second case ${\eta _2} = \frac{{2{T_1} - 2{T_2}}}{{2{T_1}}}$$ = \frac{{{T_1} - {T_2}}}{{{T_1}}} = \eta $
In second case ${\eta _2} = \frac{{2{T_1} - 2{T_2}}}{{2{T_1}}}$$ = \frac{{{T_1} - {T_2}}}{{{T_1}}} = \eta $
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
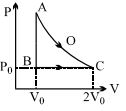
- 2An ideal gas is taken from point $A$ to point $C$ on $P-V$ diagram through two process $AOC$ and $ABC$ as shown in the figure. Process $AOC$ is isothermalView Solution
- 3$PV$ curve for the process whose $VT$ curve isView Solution
- 4A mono atomic gas is supplied the heat $Q$ very slowly keeping the pressure constant. The work done by the gas will beView Solution
- 5A diatomic ideal gas is compressed adiabatically to $\frac{1}{32}$ of its initial volume. If the initial temperature of the gas is $T_1$ (in Kelvin) and the final temperature is $a T_1$, the value of $a$ isView Solution
- 6One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $8.0 \,m ^{3}$ to $1.0 \,m ^{3}$.
Step $2$ Then expanded isothermally at temperature $T_{1}$ to volume $10.0 \,m ^{3}$.
Step $3$ Then expanded adiabatically to volume $80.0 \,m ^{3}$.
Step $4$ Then compressed isothermally at temperature $T_{2}$ to volume $8.0 \,m ^{3}$.
Then, $T_{1} / T_{2}$ is
- 7The pressure of an ideal gas varies with volume as $P = \alpha V,$ where $\alpha $ is a constant. One mole of the gas is allowed to undergo expansion such that its volume becomes $'m'$ times its initial volume. The work done by the gas in the process isView Solution
- 8Helium gas goes through a cycle $ABCDA$ ( consisting of two isochoric and isobaric lines) as shown in figure Efficiency of this cycle is nearly ....... $\%$ (Assume the gas to be close to ideal gas)View Solution
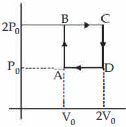
- 9View SolutionWhen you make ice cubes, the entropy of water
- 10A Carnot engine operates between ${227^o}C$ and ${27^o}C.$ Efficiency of the engine will beView Solution
