A Carnot freezer takes heat from water at $0\,^oC$ inside it and rejects it to the room at a temperature of $27\,^oC$. The latent heat of ice is $336 \times 10^3\, J\,kg^{-1}$. lf $5\, kg$ of water at $0\,^oC$ is converted into ice at $0\,^oC$ by the freezer, then the energy consumed by the freezer is close to
JEE MAIN 2016, Diffcult
$\Delta H = mL = 5 \times 336 \times {10^3} = {Q_{sink}}$
$\frac{{{Q_{sink}}}}{{{Q_{source}}}} - \frac{{{T_{sink}}}}{{{T_{source}}}}$
$\therefore {Q_{source}} = \frac{{{T_{source}}}}{{{T_{sink}}}} \times {Q_{sink}}$
Energy consumed by freezer
$\therefore {W_{output}} = {Q_{source}} - {Q_{sink}}$
$ = {Q_{sink}}\left( {\frac{{{T_{source}}}}{{{T_{sink}}}} - 1} \right)$
$Given:\,{T_{source}} = {27^ \circ }C + 273 = 300K,$
${T_{sink}} = {0^ \circ }C + 273 = 273\,K$
${W_{output}} = 5 \times 336 \times {10^3}\left( {\frac{{300}}{{273}} - 1} \right) = 1.67 \times {10^5}J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
- 2An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?View Solution
- 3View SolutionWhich one is the correct option for the two different thermodynamic processes ?
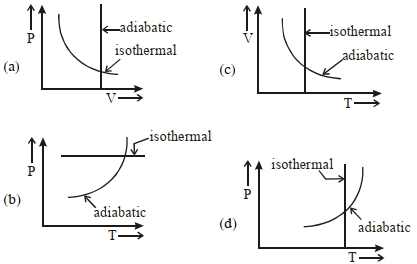
- 4The temperature inside and outside a refrigerator are $273 \,K$ and $300 \,K$ respectively. Assuming that the refrigerator cycle is reversible, for every joule of work done, the heat delivered to the surrounding will be nearly ........ $J$View Solution
- 5During an adiabatic compression, $830\, J$ of work is done on $2\, moles$ of a diatomic ideal gas to reduce its volume by $50\%$. The change in its temperahture is nearly..... $K$ $(R\, = 8.3\, J\,K^{-1}\, mol^{-1} )$View Solution
- 6The pressure and volume of a gas are changed as shown in the $P-V$ diagram in this figure. The temperature of the gas will ........View Solution
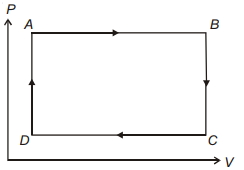
- 7Work done by a Carnot engine operating between temperatures $127^{\circ}\,C$ and $27^{\circ}\,C$ is $2\,kJ$. The amount of heat transferred to the engine by the reservoir is $........\,kJ$View Solution
- 8For an isothermal expansion of a perfect gas, the value of $\frac{{\Delta P}}{P}$ is equalView Solution
- 9Heat is supplied to a diatomic gas at constant pressure. The ratio of $\Delta Q\,:\,\Delta U\,:\,\Delta W$ isView Solution
- 10Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is trueView Solution
