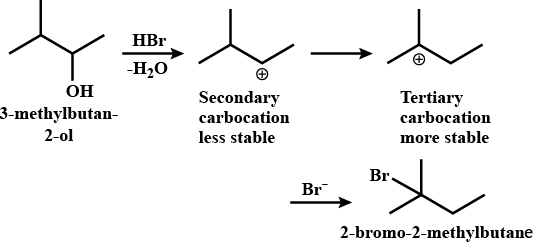
When $3-$methylbutan$-2-$ol is treated with $\ce{HBr},$ the following reaction takes place. What are the correct steps for formation of the above product?
The reaction $($along with mechanism$)$ for the conversion of $3-$methylbutan$-2-$ol to $2-$bromo$-2-$methylbutane is as given below.

Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following compounds will react with sodium hydroxide solution in water?
- 2View SolutionWhich is the only alcohol that can be prepared by the indirect hydration of alken?
- 3Isopropyl benzene on air oxidation in the presence of dilute acid gives$:$View Solution
- 4Baeyers reagent is $\ce{KMnO_4}$ in which medium?View Solution
- 5View SolutionIUPAC name of phenol is -
- 6View SolutionWhich of the following is a primary alcohol?
- 7Phenol on reaction with $NH_3$ gives:View Solution
- 8View SolutionMolecular formula of ethanol is?
- 9View SolutionMark the correct statement:
- 10View SolutionThe other name for Syngas is:
