Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionMolecular formula of ethanol is?
- 2IUPAC name of the given compound is:View Solution
$\text{H}_3\text{C}\text{−CH}−\text{CH}_2\text{−CH}\text{−CH}−\text{CH}_2−\text{CH}_3\\ \ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \ \ \ \text{OH} \ \ \ \ \ \ \ \ \ \ \ \ \ \text{OH} \ \ \ \ \text{C}_2\text{O}_5$ - 3Consider the reaction between $\ce{C_2H_{5}O^{⊝ }}$ and dimethyl sulphate. The leaving group in this reaction is$:$View Solution
- 4View SolutionWhich is the only alcohol that can be prepared by the indirect hydration of alken?
- 5View SolutionMark the correct order of decreasing acid strength of the following compounds.
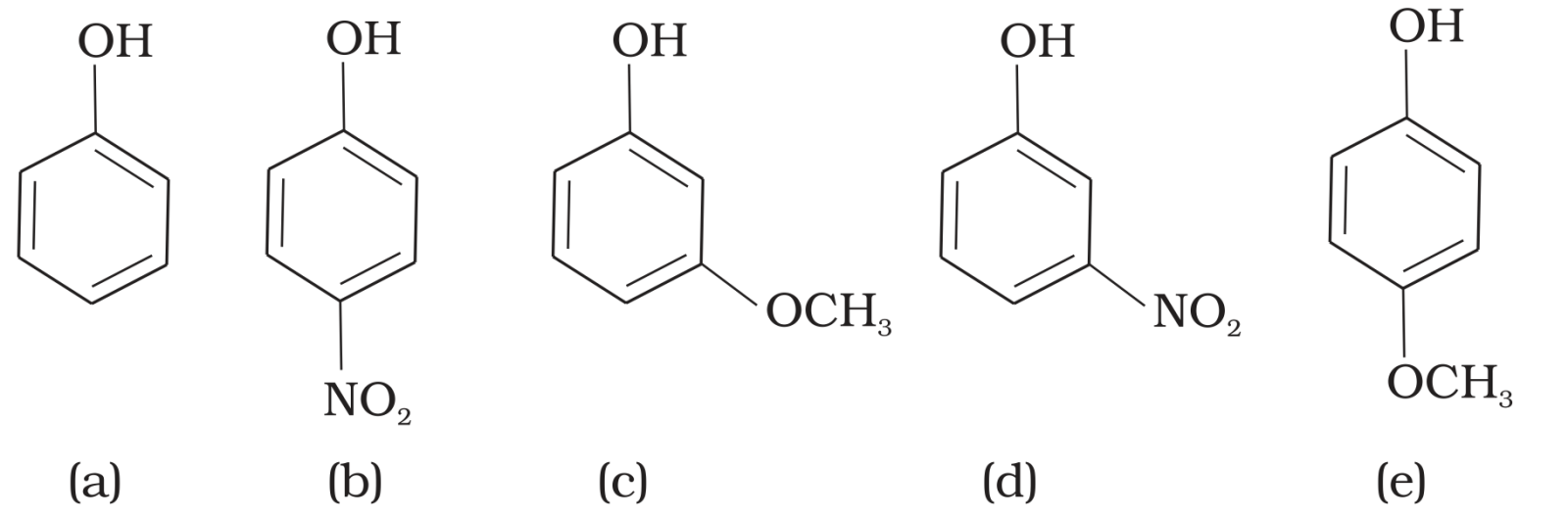
- 6View SolutionSolubility of alcohols in water is _______ to that of hydrocarbons of comparable molecular masses.
- 7View SolutionWhich of the following alcohol has highest solubility in water?
- 8Compound $\ce{C_2H_6O}$ has two isomers $X$ and $Y.$ On reaction with $HI, X$ gives alkyl iodide and water while $Y$ gives alkyl iodide and alcohol.Compounds $X$ and $Y$ are respectively:View Solution
- 9View SolutionWhich of the following reactions will yield phenol?
- 10Unlike phenol, $2,4-$dinitrophenol is soluble in sodium carbonate solution in water because?View Solution
