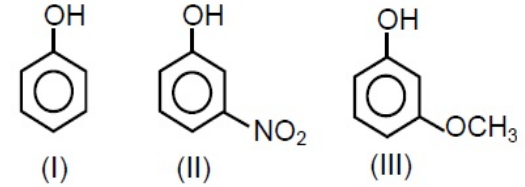
Which of the following compounds will react with sodium hydroxide solution in water?
Phenol being more acidic reacts with sodium hydroxide solution in water to give sodium phenoxide which is resonance stabilized. Alcohols are very weak acids. $\ce{C_6H_5OH + NaOH \rightarrow C_6H_5ONa + H_2O}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following represents monohydric alcohol?
- 2Unlike phenol, $2,4-$dinitrophenol is soluble in sodium carbonate solution in water because?View Solution
- 3View SolutionCorrect order of acidic strength.
- 4View SolutionAn ester can be prepared by the reaction of:
- 5Which of the following is produced during the following reaction? $\text{CO(g)} + \text{H}_2\text{(g)}\xrightarrow[\text{ZnO}\text{Cr}_2\text{O}_3]{575\text{k}}$View Solution
- 6View SolutionTrivial name of methanol is _________.
- 7View SolutionThe nature of alcoholic group in
- 8View SolutionWhich of the following phenols has lowest solubility in water?
- 9View SolutionSolubility of alcohols in water is _______ to that of hydrocarbons of comparable molecular masses.
- 10Among ethanol, dimethyl ether, methanol and propanal, the isomers are$:$View Solution
