When a gas expands adiabatically
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A carnot engine has the same efficiency between $800 K$ to $500 K$ and $x\, K$ to $600 K.$ The value of $x$ is ...... $K$View Solution
- 2A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
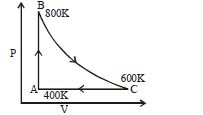
- 3One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
- 4A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
- 5The pressure $P_{1}$ and density $d_{1}$ of diatomic gas $\left(\gamma=\frac{7}{5}\right)$ changes suddenly to $P _{2}\left(> P _{1}\right)$ and $d _{2}$ respectively during an adiabatic process. The temperature of the gas increases and becomes $......$ times of its initial temperature.$\left(\right.$ given $\left.\frac{ d _{2}}{ d _{1}}=32\right)$View Solution
- 6If $\gamma = 2.5$ and volume is equal to $\frac{1}{8}$ times to the initial volume then pressure $P' $ is equal to (Initial pressure $= P$)View Solution
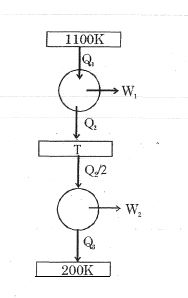
- 7Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
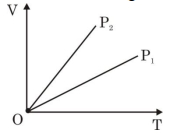
- 8View SolutionThe given figure represents two isobaric processes for the same mass of an ideal gas, then
- 9The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 10A Container having $1$ mole of a gas at a temperature $27°C$ has a movable piston which maintains at constant pressure in container of $1 \,atm.$ The gas is compressed until temperature becomes $127°C$. The work done is ........ $J$ ($C_P$ for gas is $7.03\, cal/mol-K)$View Solution
