A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The pressure and density of a diatomic gas $\gamma = 7/5$ change adiabatically from $(P, d)$ to $(P', d').$ If $\frac{{d'}}{d} = 32,$ then $\frac{{P'}}{P}$should beView Solution
- 2The heat capacity of one mole an ideal is found to be $C_V=3 R(1+a R T) / 2$, where $a$ is a constant. The equation obeyed by this gas during a reversible adiabatic expansion isView Solution
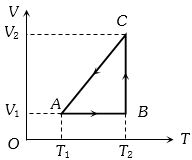
- 3A cyclic process for $1\, mole$ of an ideal gas is shown in figure in the $V-T,$ diagram. The work done in $AB, BC$ and $CA$ respectivelyView Solution
- 4Given below are two statements : one is labelled as Assertion A and the other is labelled as Reason $R$. Assertion A : Efficiency of a reversible heat engine will be highest at $-273^{\circ} C$ temperature of cold reservoir.View Solution
Reason $R$ : The efficiency of Carnot's engine depends not only on temperature of cold reservoir but it depends on the temperature of hot reservoir too and is given as $\eta=\left(1-\frac{ T _2}{ T _1}\right)$.
In the light of the above statements, choose the correct answer from the options given below
- 5$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
- 6A gas expands under constant pressure $P$ from volume ${V_1}$ to ${V_2}$. The work done by the gas isView Solution
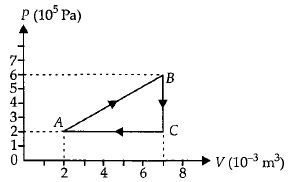
- 7A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
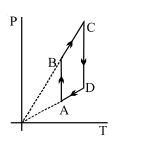
- 8View SolutionPressure versus temperature graph of an ideal gas is shown in figure
- 9A sample of an ideal gas is contained in a cylinder. The volume of the gas is suddenly decreased. A student makes the following statements to explain the change in pressure of the gasView Solution
$I.$ The average kinetic energy of the gas atoms increases
$II.$ The atoms of the gas hit the walls of the cylinder more frequently
$III.$ Temperature of the gas remains unchanged
Which of these statements is true? - 10A sample of an ideal gas undergoes an isothermal expansion. If $d Q, d U$ and $d W$ represent the amount of heat supplied, the change in internal energy and the work done respectively, thenView Solution
