When air is filled in the balloon, the pressure and volume both increases while temperature does not change. Here Boyle's law is not obeyed because
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
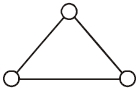
- 1Consider a gas of triatomic molecules. The molecules are assumed to the triangular and made of massless rigid rods whose vertices are occupied by atoms. The internal energy of a mole of the gas at temperature $T$ is $......RT$View Solution
- 2Air is filled at $60^o C$ in a vessel of open mouth. The vessel is heated to a temperature $T$ so that $1/4^{th}$ part of air escapes. Assuming the volume of the vessel remaining constant, the value of $T$ is ....... $^oC$View Solution
- 3View SolutionIn thermal equilibrium, the average velocity of gas molecules is
- 4A soap bubble of radius $r$ has monoatomic ideal- gas inside. The gas is heated in such a manner that bubble remains in mechanical equillibrium. Assuming that the soap material of the bubble has no heat capacity, the molar heat capacity of the gas in the process will beView Solution
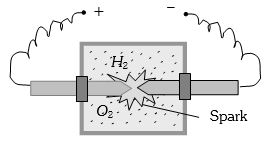
(Neglect atmospheric pressure) - 5A vessel contains $14 \,gm \,(7 $ moles) of hydrogen and $96\, gm$ ($9$ moles) of oxygen at $STP.$ Chemical reaction is induced by passing electric spark in the vessel till one of the gases is consumed. The temperature is brought back to it's starting value $273 K.$ The pressure in the vessel is ...... $atm$View Solution
- 6If the $r.m.s.$ velocity of a gas at a given temperature (Kelvin scale) is $300\, m/sec$. What will be the $r.m.s.$ velocity of a gas having twice the molecular weight and half the temperature on Kelvin scale ....... $m/sec$View Solution
- 7View SolutionAt constant volume, for different diatomic gases the molar specific heat is
- 8One mole of an ideal monatomic gas undergoes a process described by the equation $PV^3 =$ constant. The heat capacity of the gas during this process isView Solution
- 9The Mean Free Path $\ell$ for a gas molecule depends upon diameter, $d$ of the molecule asView Solution
- 10Find the approx. number of molecules contained in a vessel of volume $7$ litres at $0^oC$ at $1.3 \times 10^5$ pascalView Solution
