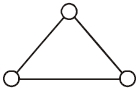
Consider a gas of triatomic molecules. The molecules are assumed to the triangular and made of massless rigid rods whose vertices are occupied by atoms. The internal energy of a mole of the gas at temperature $T$ is $......RT$

JEE MAIN 2020, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$2$ mole of $N_2$ gas at $27\,^oC$ is mixed with $1$ mole $He$ gas at $-73\,^oC$. Find $T_{mix}$ of the mixtureView Solution
- 2Two identical adiabatic vessels are filled with oxygen at pressure $P_1$ and $P_2 (P_1 > P_2).$ The vessels are interconnected with each other by a nonconducting pipe. If $U_{01}$ and $U_{02}$ denote initial internal energy of oxygen in first and second vessel respectively and $U_{f_1}$ and $U_{f_2}$ denote final internal energy values, than :View Solution
- 3For a gas the difference between the two specific heats is $4150\, J/kg\, K.$ What is the specific heats at constant volume of gas if the ratio of specific heat is $1.4$View Solution
- 4A gas mixture consists of molecules of type $1, 2$ and $3$, with molar masses ${m_1} > {m_2} > {m_3}.$ ${V_{rms}}$ and $\overline K $ are the $r.m.s.$ speed and average kinetic energy of the gases. Which of the following is trueView Solution
- 5The equation for an ideal gas is $PV = RT,$ where $V$ represents the volume ofView Solution
- 6The total kinetic energy of translatory motion of all the molecules of $5$ litres of nitrogen exerting a pressure $P$ is $3000 \,\,J$.View Solution
- 7When $2 \,\,gms$ of a gas are introduced into an evacuated flask kept at $25\,^0C$ the pressure is found to be one atmosphere. If $3 \,\,gms$ of another gas added to the same flask the pressure becomes $1.5$ atmospheres. The ratio of the molecular weights of these gases will beView Solution
- 8$3\,moles$ of an ideal gas at a temperature of $27^{\circ}\,C$ are mixed with $2\,moles$ of an ideal gas at a temperature $227^{\circ}\,C$, determine the equilibrium temperature (${}^o C$) of the mixture, assuming no loss of energy.View Solution
- 9For the $P-V$ diagram given for an ideal gas, out of the following which one correctly represents the $T-P$ diagram ?View Solution
- 10If the pressure of an ideal gas contained in a closed vessel is increased by $0.5\%,$ the increase in temperature is $2K.$ The initial temperature of the gas is ...... $^oC$View Solution
