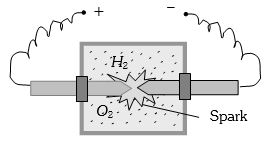
A vessel contains $14 \,gm \,(7 $ moles) of hydrogen and $96\, gm$ ($9$ moles) of oxygen at $STP.$ Chemical reaction is induced by passing electric spark in the vessel till one of the gases is consumed. The temperature is brought back to it's starting value $273 K.$ The pressure in the vessel is ...... $atm$

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA triatomic, diatomic and monatomic gas is supplied same amount of heat at constant pressure, then
- 2The change in the magnitude of the volume of an ideal gas when a small additional pressure $\Delta P$ is applied at a constant temperature, is the same as the change when the temperature is reduced by a small quantity $\Delta T$ at constant pressure. The initial temperature and pressure of the gas were $300\, K$ and $2\; atm$ respectively. If $|\Delta T|=C|\Delta P|$ then value of $C$ in $(K / a t m)$ is......View Solution
- 3At $NTP,$ sample of equal volume of chlorine and oxygen is taken. Now ratio of No. of moleculesView Solution
- 4View SolutionOn absolute temperature, the kinetic energy of the molecules
- 5At $0°C $ the density of a fixed mass of a gas divided by pressure is $x.$ At $100°C,$ the ratio will beView Solution
- 6$Assertion :$ The root mean square and most probable speeds of the molecules in a gas are the same.View Solution
$Reason :$ The Maxwell distribution for the speed of molecules in a gas in symmetrical. - 7If pressure of $C{O_2}$ (real gas) in a container is given by $P = \frac{{RT}}{{2V - b}} - \frac{a}{{4{b^2}}}$ then mass of the gas in container is ...... $gm$View Solution
- 8A sealed container with negligible coefficient of volumetric expansion contains helium (a monoatomic gas). When it is heated from $300\, K$ to $600\, K,$ the average $K.E.$ of helium atoms isView Solution
- 9The specific heat of a gas at constant volume is $21.2\, J/mole/°C.$ If the temperature is increased by $1°C$ keeping the volume constant, the change in its internal energy will be ...... $J$View Solution
- 10For a gas, the difference between the two specific heats is $4150\, J\, kg^{-1}\, K^{-1}$ and the ratio of the two specific heats is $1.4$. What is the specific heat of the gas at constant volume in units of $J\, kg^{-1}\, K^{-1}$ ?View Solution
