If the collision frequency of hydrogen molecules in a closed chamber at $27^{\circ} \mathrm{C}$ is $\mathrm{Z}$, then the collision frequency of the same system at $127^{\circ} \mathrm{C}$ is :
JEE MAIN 2024, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The $rms$ speeds of the molecules of Hydrogen, Oxygen and Carbondioxide at the same temperature are ${V}_{{H}}, {V}_{0}$ and ${V}_{{C}}$ respectively thenView Solution
- 2If the $r.m.s.$ velocity of a gas at a given temperature (Kelvin scale) is $300\, m/sec$. What will be the $r.m.s.$ velocity of a gas having twice the molecular weight and half the temperature on Kelvin scale ....... $m/sec$View Solution
- 3$\mathrm{N}$ moles of a polyatomic gas $(f=6)$ must be mixed with two moles of a monoatomic gas so that the mixture behaves as a diatomic gas. The value of $\mathrm{N}$ is:View Solution
- 4The pressure and temperature of two different gases is $P$ and $T$ having the volume $V$ for each. They are mixed keeping the same volume and temperature, the pressure of the mixture will beView Solution
- 5A closed hollow insulated cylinder is filled with gas at ${0^o}C$ and also contains an insulated piston of negligible weight and negligible thickness at the middle point. The gas on one side of the piston is heated to ${100^o}C.$ If the piston moves $5\,cm,$ the length of the hollow cylinder is ..... $cm$View Solution
- 6View SolutionOn the basis of kinetic theory of gases, the gas exerts pressure because its molecules:
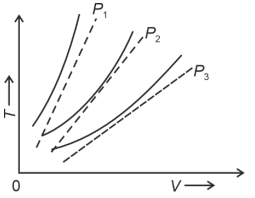
- 7The following graph represents the $T-V$ curves of an ideal gas (where $T$ is the temperature and $V$ the volume) at three pressures $P_1, P_2$ and $P_3$ compared with those of Charles's law represented as dotted lines.View Solution
Then the correct relation is:
- 8If the root mean square velocity of the molecules of hydrogen at $NTP$ is $1.84\, km/s$. Calculate the root mean square velocity of oxygen molecule at $NTP$, molecular weight of hydrogen and oxygen are $2$ and $32$ respectively ....... $km/sec$View Solution
- 9According to kinetic theory of gases,View Solution
$A$. The motion of the gas molecules freezes at $0^{\circ} C$
$B$. The mean free path of gas molecules decreases if the density of molecules is increased.
$C$. The mean free path of gas molecules increases if temperature is increased keeping pressure constant.
$D$. Average kinetic energy per molecule per degree of freedom is $\frac{3}{2} k_{B} T$ (for monoatomic gases)
Choose the most appropriate answer from the options given below
- 10In the absence of intermolecular force of attraction, the observed pressure $P$ will beView Solution
