When volume of system is increased two times and temperature is decreased half of its initial temperature, then pressure becomes ...... times
AIEEE 2002, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two moles of helium are mixed with $n$ with moles of hydrogen. If $\frac{{{C_P}}}{{{C_V}}}\, = \,\frac{3}{2}$ for the mixture, then the value of $n$ isView Solution
- 2At $NTP,$ sample of equal volume of chlorine and oxygen is taken. Now ratio of No. of moleculesView Solution
- 3View SolutionIf a Vander-Waal's gas expands freely, then final temperature is
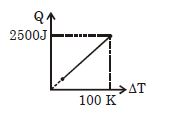
- 4One mole of a gas mixture is heated under constant pressure, and heat supplied $Q$ is plotted against temperature difference acquired. Find the approximate value of $\gamma $ for mixtureView Solution
- 5View SolutionThe specific heat of an ideal gas is
- 6Inside a cylinder having insulating walls and closed at ends is a movable piston, which divides the cylinder into two compartments. On one side of the piston is a mass $m$ of a gas and on the other side a mass $2 m$ of the same gas. What fraction of volume of the cylinder will be occupied by the larger mass of the gas when the piston is in equilibrium $?$ Consider that the movable piston is conducting so that the temperature is the same throughoutView Solution
- 7View SolutionThe molecules of air in the room that you are sitting are all experiencing the force of gravity tending to bring them down. The molecules are also frequently and randomly undergoing collisions, which tend to oppose the effect of fall under gravity. The density of air is nearly uniform throughout the room because
- 8View SolutionThe time average of the kinetic energy of one molecule of a gas taken over a long period of time
- 9Two identical bodies are made of a material for which the heat capacity increases with temperature. One of these is held at a temperature of $100^{\circ} C$, while the other one is kept at $0^{\circ} C$. If the two are brought into contact, then assuming no heat loss to the environment, the final temperature that they will reach isView Solution
- 10What is the ratio of specific heats of constant pressure and constant volume for $N{H_3}$View Solution
