Which is the correct statement
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
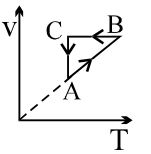
- 1View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
- 2Unit mass of a liquid with volume ${V_1}$ is completely changed into a gas of volume ${V_2}$ at a constant external pressure $P$ and temperature $T.$ If the latent heat of evaporation for the given mass is $L,$ then the increase in the internal energy of the system isView Solution
- 3Determine coefficient of performance of given temperature limit.View Solution
$T_{1}=27^{\circ} C$ [outside fridge]
$T_{2}=-23^{\circ} C$ [inside fridge]
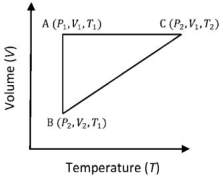
- 4A reversible cyclic process for an ideal gas is shown below. Here, $P , V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$ $(B)$ $W _{ BC }= P _2\left( V _2- V _1\right)$ and $q _{ BC }= H _{ AC }$ $(C)$ $\Delta H _{ CA }<\Delta U _{ CA }$ and $q _{ AC }=\Delta U _{ BC }$ $(D)$ $q_{B C}=\Delta H_{A C}$ and $\Delta H_{C A}>\Delta U_{C A}$
- 5View SolutionIn which of the following processes, heat is neither absorbed nor released by a system ?
- 6Gas obey $P^2V =$ constant. The initial temperature and volume are $T_0$ and $V_0$. If gas expands to volume $2V_0$, the final temp isView Solution
- 7View SolutionIn a thermodynamic system working substance is ideal gas, its internal energy is in the form of
- 8Determine coefficient of performance of given temperature limit.View Solution
$T_{1}=27^{\circ} C$ [outside fridge]
$T_{2}=-23^{\circ} C$ [inside fridge]
- 9A heat engine is involved with exchange of heat of $1915\, J,$ $-40\, J ,+125\, J$ and $-Q\,J$, during one cycle achieving an efficiency of $50.0 \%$. The value of $Q$ is.......$J$View Solution
- 10An ideal gas is expanding such that $\mathrm{PT}^2=$ constant. The coefficient of volume expansion of the gas isView Solution
