Gas obey $P^2V =$ constant. The initial temperature and volume are $T_0$ and $V_0$. If gas expands to volume $2V_0$, the final temp is
Medium
$\mathrm{P}^{2} \mathrm{V}=\mathrm{constant}$
$\left(\frac{\mathrm{nRT}}{\mathrm{V}}\right)^{2} \mathrm{V}=\mathrm{constant}$
$\frac{\mathrm{T}^{2}}{\mathrm{V}}=\mathrm{constant}$
$\frac{\mathrm{T}_{1}^{2}}{\mathrm{V}_{1}}=\frac{\mathrm{T}_{2}^{2}}{\mathrm{V}_{2}} \quad \Rightarrow \quad \mathrm{T}_{2}^{2}=\frac{\mathrm{V}_{2}}{\mathrm{V}_{1}} \mathrm{T}_{1}^{2}=2 \mathrm{T}_{0}^{2}$
$\Rightarrow \mathrm{T}_{2}=\sqrt{2} \mathrm{T}_{0}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
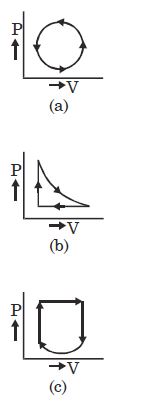
- 1View SolutionWhat is the nature of change in internal energy in the following three thermodynamical processes shown in figure
- 2The volume of an ideal gas is $1$ litre and its pressure is equal to $72cm$ of mercury column. The volume of gas is made $900\, cm^3$ by compressing it isothermally. The stress of the gas will be ...... $cm$ (mercury)View Solution
- 3The gas law $\frac{{PV}}{T} = $ constant is true forView Solution
- 4An engine runs between a reservoir at temperature $200 \,K$ and a hot body which is initially at temperature of $600 \,K$. If the hot body cools down to a temperature of $400 \,K$ in the process, then the maximum amount of work that the engine can do (while working in a cycle) is (the heat capacity of the hot body is $1 \,J / K )$View Solution
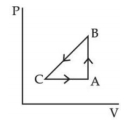
- 5A sample of an ideal gas is taken through the cyclic process $ABCA$ as shown in figure. It absorbs, $40\,J$ of heat during the part $A B$, no heat during $BC$ and rejects $60\,J$ of heat during $CA$. $A$ work $50\,J$ is done on the gas during the part $BC$. The internal energy of the gas at $A$ is $1560\,J$. The work done by the gas during the part $CA$ is.............$J$View Solution
- 6A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.View Solution
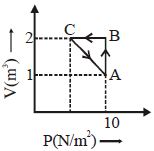
- 7An ideal gas is taken through the cycle $A \to B \to C \to A,$ as shown in the figure. What is the change in internal energy ...... $J$.View Solution
- 8In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
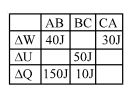
- 9A student records $\Delta Q, \Delta U \& \Delta W$ for a thermodynamic cycle $A$ $\rightarrow$ $B \rightarrow C \rightarrow A$. Certain entries are missing. Find correct entry in following options.View Solution
- 10Find the amount of work done to increase the temperature of one mole of an ideal gas by $30^o\ C$ if it is expanding under the condition .... $J$View Solution
$V\propto {T^{\frac{2}{3}}}$ $[R = 1.99\ cal/mol-K]$
