Which of the following alcohol does not give a stable compound on dehydration?
Alcohols undergo dehydration $($removal of water$)$ to form an alkene.
To form alkene, we need at least two carbon atoms. But, methanol $\ce{(CH_3OH)}$ has only one carbon atom. So, it does not give a stable compound on dehydration.
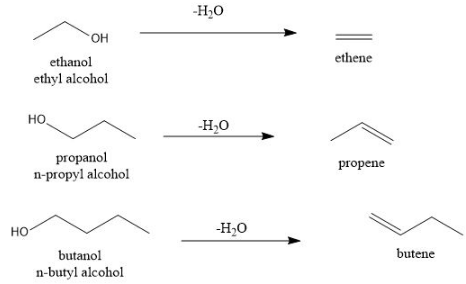
To form alkene, we need at least two carbon atoms. But, methanol $\ce{(CH_3OH)}$ has only one carbon atom. So, it does not give a stable compound on dehydration.

Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
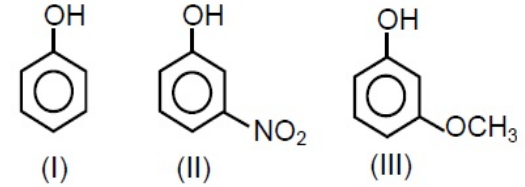
- 1View SolutionCorrect order of acidic strength.
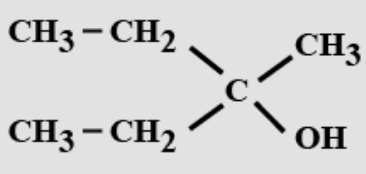
- 2View SolutionThe IUPAC name is:
- 3Unlike phenol, $2,4-$dinitrophenol is soluble in sodium carbonate solution in water because?View Solution
- 4View SolutionThe process of converting alkyl halides into alcohols involves_____________.
- 5Write the $\ce{IUPAC}$ name of the given compound:$\text{HO}−\text{CH}_2−\text{CH}−\text{CH}_2−\text{OH}\\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ ∣ \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{CH}_3$View Solution
- 6View SolutionWhich of the following has a higher boiling point?
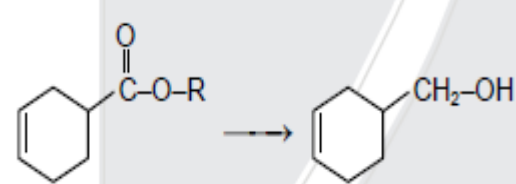
- 7View SolutionWhich reagent is suitable for this conversion?
- 8Give IUPAC name of the compound given below.View Solution
$\text{CH}_3-\text{CH}-\text{CH}_2-\text{CH}_2-\text{CH}-\text{CH}_3 \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{Cl} \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{CH}$ - 9View SolutionVapours of an alcohol X when passed over hot reduced copper, produce an alkene, the alcohol is:
- 10Carbinol is a trivial name of$:$View Solution
