Unlike phenol, $2,4-$dinitrophenol is soluble in sodium carbonate solution in water because?
Presence of two electron withdrawing $\ce{−No_2}$ groups in the ring makes $2,4-$dinitrophenol a stronger acid than phenol.
Hence it react with aqueous $\ce{Na_2CO_3}$ solution to form sodium salt thus making it soluble in $\ce{Na_2CO_3}.$
Hence it react with aqueous $\ce{Na_2CO_3}$ solution to form sodium salt thus making it soluble in $\ce{Na_2CO_3}.$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The number of carbon atoms present in a molecule of simple ether is$:$View Solution
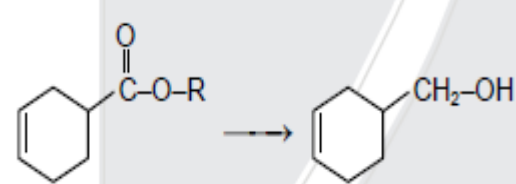
- 2View SolutionWhich reagent is suitable for this conversion?
- 3View SolutionDestructive distillation of wood in the laboratory will result in the formation of:
- 4View SolutionLower alcohols are highly soluble in water due to _____.
- 5View SolutionThe decreasing order of boiling points of the following alcohols is?
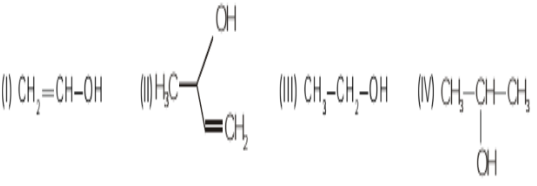
- 6View SolutionWhich of the following represents monohydric alcohol?
- 7View SolutionWhat is the hybridisation of carbon and oxygen in electronic structure of ether?
- 8View SolutionWhich of the following alcohol has the highest boiling point?
- 9View SolutionMethanol is now prepared from:
- 10View SolutionThe correct reactivity order of alcohols towards H−X will be: