Which of the following alcohols is dehydrated most easily with conc. $\ce{H_2SO_4}$?
- $\ce{p−CH_3OC_6H_4CH(OH)CH_{3}}$
$\rightarrow$ alkene is an elimination reaction that goes via carbocation formation.
Higher is the stability of carbocation more easily it can be dehydrated.
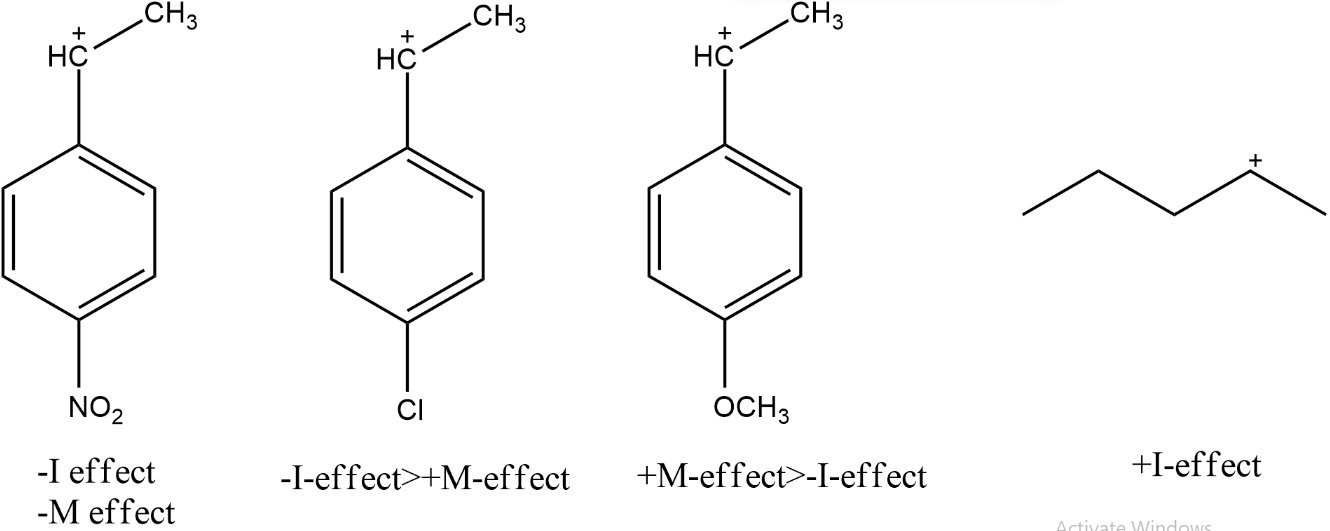
The corresponding carbocations formed by given molecules are shown in the figure.
Due to the $--$Inductive effect $(-I)$and $--$Mesomeric effect $(-M)$ of $NO_2$ group, it will decrease the electron density on benzene and will, therefore, destabilise the carbocation the most and makes it difficult to dehydrate.
In case of $\ce{p−ClC_6H_4CH(OH)CH_3}$, the Cl has stronger $-I$ effect $($due to high electronegativity$)$ than its +M effect (due to a lone pair of electrons) thus will destabilise the carbocation and unfavour dehydration.
In case of $\ce{p−CH_3OC_{6}H_4CH(OH)CH_3}$, the $CH_3O$ has the stronger $+M$ effect due to a lone pair of electrons on $O$ than its $-I$ effect and will stabilise the carbocation by increasing the electron density in the benzene ring and will favour dehydration.
$\ce{C_3H_5CH(OH)CH_3}$ has $+I$ effect that stabilises the carbocation but its impact is lower than the $+-$mesomeric effect of the methoxy group.
The alcohol that is dehydrated most easily with conc. $\ce{H_{2}SO_4}$ is $\ce{p−CH_3OC_{6}H_4CH(OH)CH_3}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe correct IUPAC name of the compound is?

- 2View SolutionWith ethoxyethane which compound is miscible to almost the same extent?
- 3The number of carbon atoms present in a molecule of simple ether is$:$View Solution
- 4View SolutionOn reacting with grignard reagent acetone gives?
- 5View SolutionThe IUPAC name of the following compound is:
- 6View SolutionSelect the correct order of boiling point:
- 7The high boiling point of ethanol $(78.2^\circ C)$ compared to dimethyl ether $(−23.6^\circ C),$ though both having the same molecular formula $C_2H_6O,$ is due to:View Solution
- 8Ethyl alcohol exhibits acidic character on reacting with$:$View Solution
- 9View SolutionWhich of the following alcohol on dehydration will produce a pair of isomeric alkene?
- 10View SolutionDestructive distillation of wood in the laboratory will result in the formation of:
