The high boiling point of ethanol $(78.2^\circ C)$ compared to dimethyl ether $(−23.6^\circ C),$ though both having the same molecular formula $C_2H_6O,$ is due to:
The high boiling point of ethanol $(78.2^\circ C)$ compared to dimethyl ether $(−23.6^\circ C),$ though both having the same molecular formula $C_{2}H_6O,$ is due to Hydrogen bonding.Hydrogen bonding is observed when $H$ atom is attached to more electronegative $N, F$ or $O$ atom.
Ethanol molecules are associated due to hydrogen bonding. This results in higher boiling point.
However, hydrogen bonding is not possible in dimethyl ether.
Ethanol molecules are associated due to hydrogen bonding. This results in higher boiling point.
However, hydrogen bonding is not possible in dimethyl ether.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following is used in the manufacture of ethanol?
- 2In an aqueous solution at $25^\circ C$ has twice as many $OH^-$ as pure water its $\ce{pOH}$ will be$:$View Solution
- 3View SolutionWhich compound is the most soluble in water?
- 4View SolutionMonochlorination of toluene in sunlight followed by hydrolysis with aq. NaOH yields.
- 5Reagents used for industrial preparation of methanol are$:$View Solution
- 6View SolutionMark the correct statement:
- 7View SolutionGive the IUPAC name of the above compound.
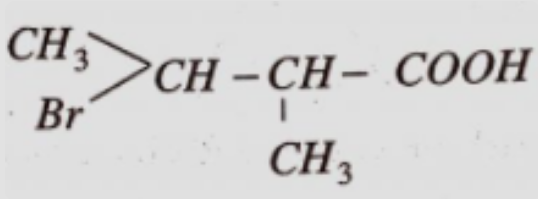
- 8View SolutionThe IUPAC name of sec. butyl alcohol is:
- 9Ethene reacts with $Y$ to produce ethanol. ethene $+ Y \rightarrow$ ethanol What is $Y$?View Solution
- 10View SolutionWhich is the best reagent to convert cyclohexanol into cyclohexene?
