Select the correct order of boiling point:
- n-butyl alcohol > n-butaraldehyde > n-pentane > diethylether.
As we know that the higher the extent of intermolecular Hydrogen bonding in a molecule, the higher its boiling point becomes.
By that logic, n-butyl alcohol will have a higher boiling point than n-butyraldehyde due to the presence of more extensive H-bonding.
Again, there is no H-bonding present in both diethyl ether and n-pentane. But as n-pentane has a higher molecular weight than that of diethyl ether, it will possess a higher boiling point.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe miscibility of ether with _______ resembles those of alcohol of the same molecular mass.
- 2View SolutionWhich of the following has a higher boiling point?
- 3IUPAC name of the given compound is:View Solution
$\text{H}_3\text{C}\text{−CH}−\text{CH}_2\text{−CH}\text{−CH}−\text{CH}_2−\text{CH}_3\\ \ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \ \ \ \text{OH} \ \ \ \ \ \ \ \ \ \ \ \ \ \text{OH} \ \ \ \ \text{C}_2\text{O}_5$ - 4View SolutionAlcohols containing only up to ________________ carbon atoms are completely miscible with water.
- 5View SolutionDehydration of the following in increasing order is:
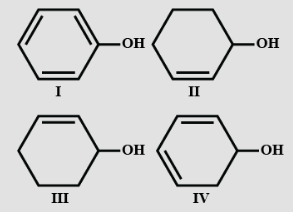
- 6Unlike phenol, $2,4-$dinitrophenol is soluble in sodium carbonate solution in water because?View Solution
- 7View SolutionWhich of the following combinations can be used for the preparation of cis vic diol?
- 8View SolutionWood spirit if known as?
- 9View SolutionWhich of the following is most acidic?
- 10$\text{CH}_3\text{−CH}\text{−CH}−\text{CH}_3\\ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \text{OH}\ \ \ \ \ \ \text{OH}$IUPAC name of the given compound is:View Solution
