Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionBy treating alkylchloride with aq. KOH produce:
- 2The $\text{IUPAC}$ name for $\ce{CH_3CH(OH)CH_2C(CH_3)_2OH}$ is$:$View Solution
- 3Ether is formed when alkyl halide is treated with sodium alkoxide. This method is known as$:$View Solution
- 4View SolutionWhich of the following species can act as the strongest base?
- 5View SolutionThe correct reactivity order of alcohols towards H−X will be:
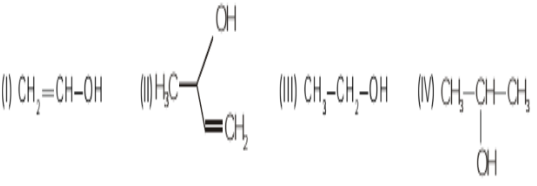
- 6View SolutionWhich of the following species show maximum volatility?
- 7Consider the reaction between $\ce{C_2H_{5}O^{⊝ }}$ and dimethyl sulphate. The leaving group in this reaction is$:$View Solution
- 8The compound which gets dissolved in water is$:$View Solution
- 9View SolutionWhich of the following is the functional group of an ether?
- 10View SolutionTrivial name of methanol is _________.
