The correct reactivity order of alcohols towards H−X will be: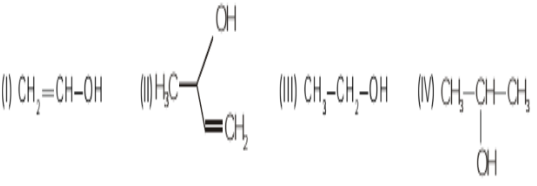

- II>IV>III>I
The correct reactivity order of alcohols towards H−X will be II>IV>III>I
Alcohol II has maximum reactivity as the carbocation formed will be stabilized by resonance with adjacent C=C double Bond.
Alcohol I has minimum reactivity as the carbocation formed will have positive charge on sp2 carbon atom.
Alcohol IV is more reactive than alcohol III because alcohol IV gives secondary carbocation whereas alcohol III gives primary carbocation.
Secondary carbocation is more stable than primary carbocation.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe decreasing order of boiling points of the following alcohols is?
- 2View SolutionThe boiling point of methanol is greater than that of methyl thiol because:
- 3IUPAC name of the compound $\text{CH}_3-\text{CH}-\text{OCH}_3 \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{CH}_3$ is ___________.View Solution
- 4The only alcohol that can be prepared by the indirect hydration of alkene is$:$View Solution
- 5View SolutionMolecular formula of ethanol is?
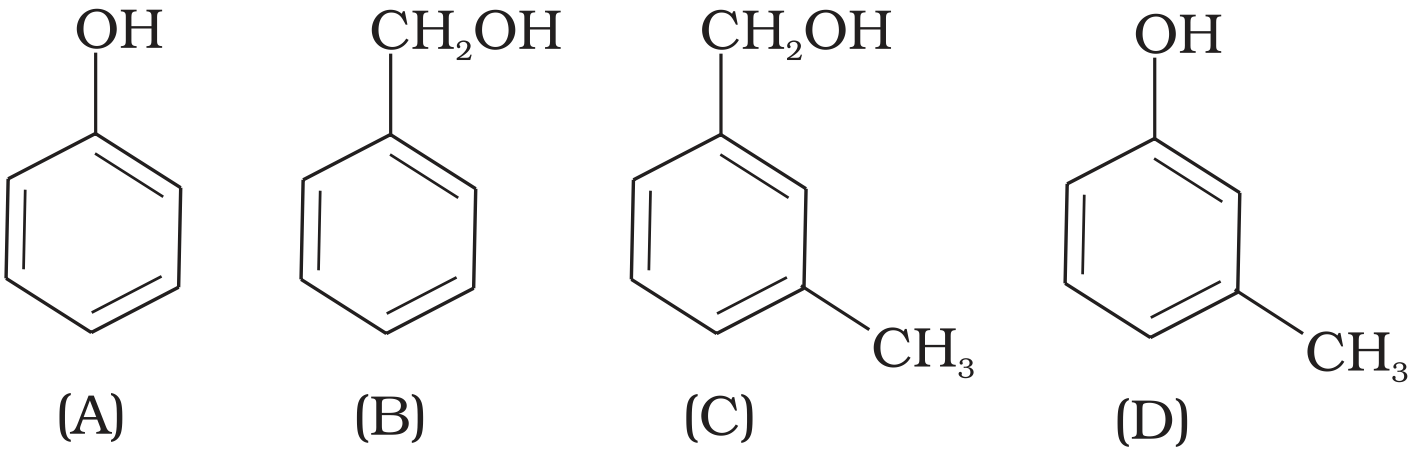
- 6View SolutionWhich of the following compounds is aromatic alcohol?
- 7View SolutionThe correct order of boiling point for primary (1), secondary (2) and tertiary (3) alcohols is:
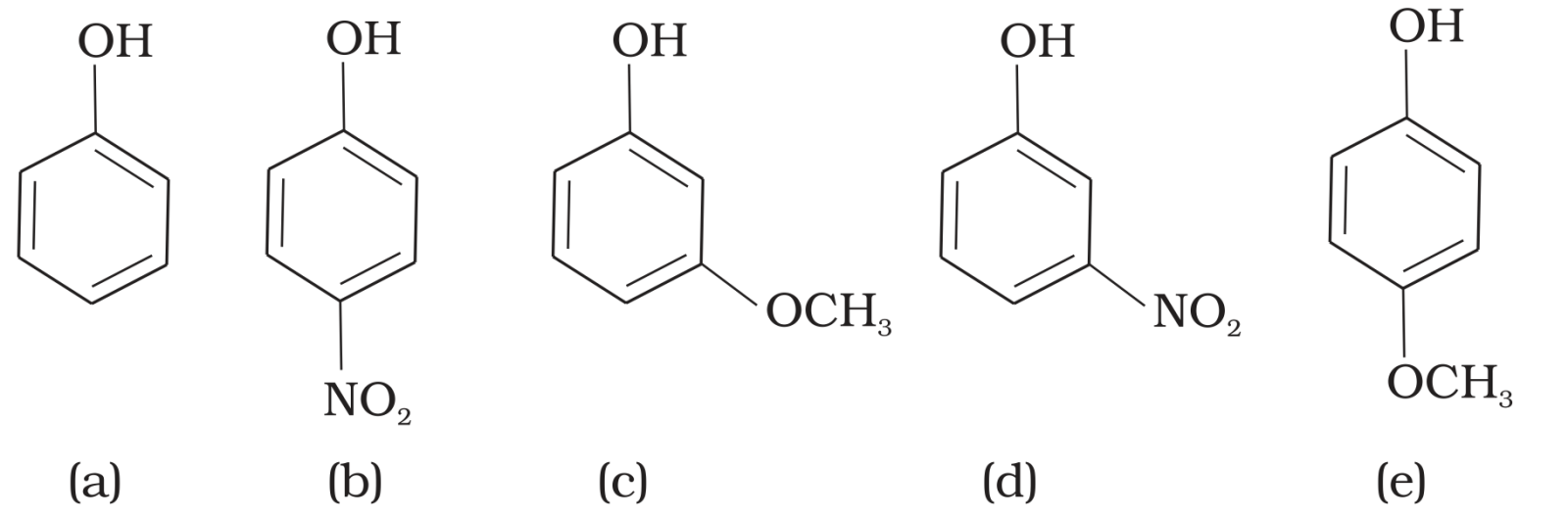
- 8View SolutionMark the correct order of decreasing acid strength of the following compounds.
- 9View SolutionWhich of the following alcohol on dehydration will produce a pair of isomeric alkene?
- 10View Solution

 is-
is-