Which of the following statement is not correct?
Phenol does not react with $\ce{Na_2CO_3}$ because it is weaker acid than carboxylic acid and thereby do not have the strength to substitute or give away its $H+$ ions to that of weak bases like sodium carbonate.
Phenol is used for the preparation of aspirin which is used as an analgesic as well as antipyretic drugs.
Phenol is more soluble in water than chlorobenzene due to formation of $H-$bond with water molecules.
$o-$nitrophenol form intramolecular $H-$bonding while $p-$nitrophenol form intermolecular $H-$bonding.
Due to this, nature $o-$nitrophenol has a lower boiling point than $p-$nitrophenol.
Phenol is used for the preparation of aspirin which is used as an analgesic as well as antipyretic drugs.
Phenol is more soluble in water than chlorobenzene due to formation of $H-$bond with water molecules.
$o-$nitrophenol form intramolecular $H-$bonding while $p-$nitrophenol form intermolecular $H-$bonding.
Due to this, nature $o-$nitrophenol has a lower boiling point than $p-$nitrophenol.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe nature of alcoholic group in
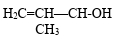
- 2View SolutionWhich reagent is suitable for this conversion?
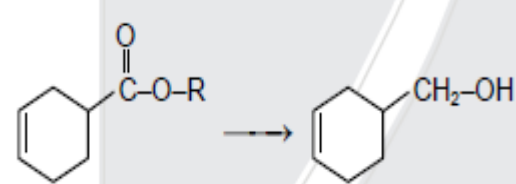
- 3What amount of bromine will be required to convert $2g$ of phenol into $2, 4, 6−$tribromophenol?View Solution
- 4Among ethanol, dimethyl ether, methanol and propanal, the isomers are$:$View Solution
- 5View SolutionThe major product obtained on acid - catalysed hydration of 2-phenylpropene is:
- 6View SolutionThe common name for propan-2-ol is:
- 7$\text{CH}_3\text{−CH}\text{−CH}−\text{CH}_3\\ \ \ \ \ \ \ \ \ \ \ \ \ | \ \ \ \ \ \ \ \ \ \ | \\ \ \ \ \ \ \ \ \ \ \text{OH}\ \ \ \ \ \ \text{OH}$IUPAC name of the given compound is:View Solution
- 8Write the $\ce{IUPAC}$ name of the given compound:$\text{HO}−\text{CH}_2−\text{CH}−\text{CH}_2−\text{OH}\\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ ∣ \\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \text{CH}_3$View Solution
- 9View SolutionPhenol is less acidic than ______________.
- 10View SolutionCommon name for methanol is:
