Work done by a system under isothermal change from a volume ${V_1}$ to ${V_2}$ for a gas which obeys Vander Waal's equation $(V - \beta n)\,\left( {P + \frac{{\alpha {n^2}}}{V}} \right) = nRT$
Diffcult
(a)According to given Vander Waal’s equation
$P = \frac{{nRT}}{{V - n\beta }} - \frac{{\alpha {n^2}}}{{{V^2}}}$
Work done, $W = \int_{{V_1}}^{{V_2}} {PdV} = nRT\int_{{V_1}}^{{V_2}} {\frac{{dV}}{{V - n\beta }}} - \alpha {n^2}\int_{{V_1}}^{{V_2}} {\frac{{dV}}{{{V^2}}}} $
$ = nRT\,\left[ {{{\log }_e}(V - n\beta )} \right]\,_{{V_1}}^{{V_2}} + \alpha {n^2}\left[ {\frac{1}{V}} \right]_{{V_1}}^{{V_2}}$
$ = nRT{\log _e}\frac{{{V_2} - n\beta }}{{{V_1} - n\beta }} + \alpha {n^2}\left( {\frac{{{V_1} - {V_2}}}{{{V_1}{V_2}}}} \right)$
$P = \frac{{nRT}}{{V - n\beta }} - \frac{{\alpha {n^2}}}{{{V^2}}}$
Work done, $W = \int_{{V_1}}^{{V_2}} {PdV} = nRT\int_{{V_1}}^{{V_2}} {\frac{{dV}}{{V - n\beta }}} - \alpha {n^2}\int_{{V_1}}^{{V_2}} {\frac{{dV}}{{{V^2}}}} $
$ = nRT\,\left[ {{{\log }_e}(V - n\beta )} \right]\,_{{V_1}}^{{V_2}} + \alpha {n^2}\left[ {\frac{1}{V}} \right]_{{V_1}}^{{V_2}}$
$ = nRT{\log _e}\frac{{{V_2} - n\beta }}{{{V_1} - n\beta }} + \alpha {n^2}\left( {\frac{{{V_1} - {V_2}}}{{{V_1}{V_2}}}} \right)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA cycle tyre bursts suddenly. This represents an
- 2One mole of an ideal monoatomic gas is heated at a constant pressure of one atmosphere from ${0^o}C$ to ${100^o}C$. Then the change in the internal energy isView Solution
- 3A gas is compressed from a volume of $2\,m^3$ to a volume of $1\, m^3$ at a constant pressure of $100\, N/m^2$. Then it is heated at constant volume by supplying $150\, J$ of energy. As a result, the internal energy of the gasView Solution
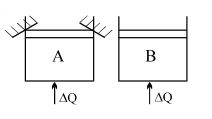
- 4Two identical vessels $A \& B$ contain equal amount of ideal monoatomic gas. The piston of $A$ is fixed but that of $B$ is free. Same amount of heat is absorbed by$A \& B$. If $B'$s internal energy increases by $100 \,\,J$ the change in internal energy of $A$ is ...... .$J$View Solution
- 5A Carnot engine operating between two reservoirs has efficiency $\frac{1}{3}$. When the temperature of cold reservoir raised by $x$, its efficiency decreases to $\frac{1}{6}$. The value of $x$, if the temperature of hot reservoir is $99^{\circ}\,C$, will be $........\,K$View Solution
- 6A perfect gas of a given mass is heated first in a small vessel and then in a large vessel, such that their volumes remain unchanged. The $P-T$ curves areView Solution
- 7Two cylinders $A$ and $B$ fitted with pistons contain equal amounts of an ideal diatomic gas at $300 K$ . The piston of $A$ is free to move while that of $B$ is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in $A$ is $30 K$ , then the rise in temperature of the gas in $B$ is ..... $K$View Solution
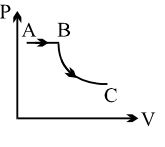
- 8View SolutionA process is shown in the diagram. Which of the following curves may represent the same process ?
- 9View SolutionEntropy of a thermodynamic system does not change when this system is used for
- 10An ideal gas follows a process described by the equation $PV ^2= C$ from the initial $\left( P _1, V _1, T _1\right)$ to final $\left(P_2, V_2, T_2\right)$ thermodynamics states, where $C$ is a constant. ThenView Solution
