One mole of an ideal monoatomic gas is heated at a constant pressure of one atmosphere from ${0^o}C$ to ${100^o}C$. Then the change in the internal energy is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Work done by a system under isothermal change from a volume ${V_1}$ to ${V_2}$ for a gas which obeys Vander Waal's equation $(V - \beta n)\,\left( {P + \frac{{\alpha {n^2}}}{V}} \right) = nRT$View Solution
- 2View SolutionAir in a cylinder is suddenly compressed by a piston, which is then maintained at the same position. With the passage of time
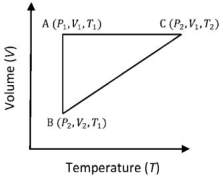
- 3A reversible cyclic process for an ideal gas is shown below. Here, $P , V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$ $(B)$ $W _{ BC }= P _2\left( V _2- V _1\right)$ and $q _{ BC }= H _{ AC }$ $(C)$ $\Delta H _{ CA }<\Delta U _{ CA }$ and $q _{ AC }=\Delta U _{ BC }$ $(D)$ $q_{B C}=\Delta H_{A C}$ and $\Delta H_{C A}>\Delta U_{C A}$
- 4A gas may expand either adiabatically or isothermally. A number of $P-V$ curves are drawn for the two processes over different range of pressure and volume. It will be found thatView Solution
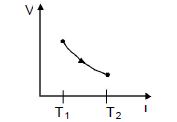
- 5View SolutionThe given figure represents two isobaric processes for the same mass of an ideal gas, then
- 6The temperature inside and outside of refrigerator are $260\, K$ and $315\, K$ respectively. Assuming that the refrigerator cycle is reversible, calculate the heat delivered to surroundings for every joule of work done.View Solution
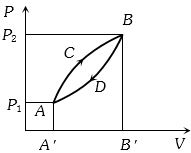
- 7A thermodynamic system is taken from state $A$ to $B$ along $ACB$ and is brought back to $A$ along $BDA$ as shown in the $PV$ diagram. The net work done during the complete cycle is given by the areaView Solution
- 8View SolutionA gas is compressed adiabatically, which one of the following statement is NOT true.
- 9If ${C_V} = 4.96cal/mole\, K$, then increase in internalenergy when temperature of $2$ moles of this gas is increased from $340 K$ to $342 K$ ....... $cal$View Solution
- 10The figure shows a process $AB$ undergone by $2$ moles of an ideal diatomic gas. The process $AB$ is in such a way that $VT =$ constant. $T_1 = 300 K $and $T_2 = 500 K$ ( $R = $ gas constant)View Solution