Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionAir in a cylinder is suddenly compressed by a piston, which is then maintained at the same position. With the passage of time
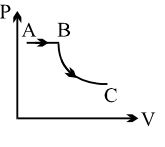
- 2View SolutionIn pressure-volume diagram given below, the isochoric, isothermal, and isobaric parts respectively, are
- 3How much work to be done in decreasing the volume of and ideal gas by an amount of $2.4 \times {10^{ - 4}}{m^3}$ at normal temperature and constant normal pressure of .......$joule$ $1 \times {10^5}N/{m^2}$View Solution
- 4Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
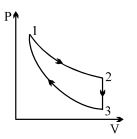
- 5An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
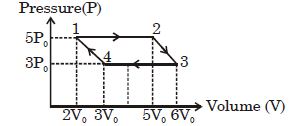
- 6Consider a carnot's cycle operating between $T_1 = 500\,K$ and $T_2 = 300\,K$ producing $1\,kJ$ of mechanical work per cycle. Find the heat transferred to the engine by the reservoirs .... $J$View Solution
- 7Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative.View Solution
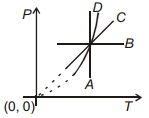
- 8A Carnot engine takes $6000 \,cal$ of heat from a reservoir at $627^{\circ} C$ and gives it to a sink at $27^{\circ} C$. The work done by the engine is ......... $kcal$View Solution
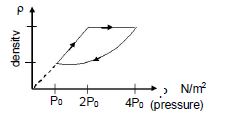
- 9An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
- 10If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution